- Home
- >
- Taiwan
- >
- Regulatory Affairs
Regulatory Affairs Services in Taiwan

Services We Offer
Life Sciences Product Registration in Taiwan
Taiwan’s life sciences sector is progressively growing, driven by a public health ecosystem with increasing healthcare investments, enhanced demand for innovative healthcare products, and growing aging population. As of 2025, Taiwan’s pharmaceutical industry is projected to grow by US $4.09 billion market, and is forecasted to reach US $4.92 bn.
The Taiwan Food and Drug Administration (TFDA), under the Ministry of Health and Welfare (MOHW), operates the regulatory framework for pharmaceuticals, medical devices, cosmetics, nutraceuticals, and veterinary products.
Taiwan is a key entry point into the Asia-Pacific market due to its regulatory affairs system following international standards like ICH, PIC/S, and GHTF. Entering this market requires proper documentation, compliance with local requirements, and working closely with a Taiwan-based license holder. A trusted regulatory affairs consultant in Taiwan can guide you through the process and help manage local regulatory needs effectively.
Regulatory Affairs Process In Taiwan
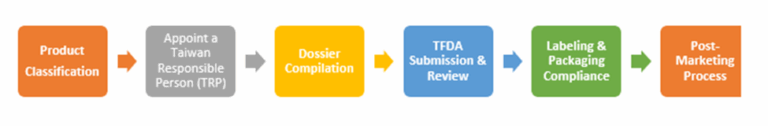
Step 1 - Product Classification
Classifying a product is necessary to determine the required documentation and regulatory approval pathway. A regulatory affairs consultant or regulatory affairs consulting company can help assess the correct product category and guide you through the appropriate regulatory steps. Examples include:
- Pharmaceuticals: New, generic, biosimilar, OTC
- Medical Devices: Class I (low risk), Class II (moderate), Class III (high risk)
- Cosmetics: General or special purpose
Step 2 - Appoint a Taiwan Responsible Person (TRP)
Step 3 - Dossier Compilation
- Administrative documents
- Technical documentation (including QSD for devices)
- Non-clinical and clinical data
- Product information in traditional Chinese
- GMP/QMS certification from a recognized country
Step 4 - TFDA Submission and Review
- Medical Devices: 2–6 months (Class I–III)
- Pharmaceuticals: 6–12 months
- Cosmetics: 2–4 months
Step 5 - Labeling and Packaging Compliance
- Localized labeling should be in traditional Chinese (mandatory).
- Cosmetic and health food labels must meet font size, allergen information, and ingredient placement regulations.
Step 6 - Product Approval & Post-Market Obligations
- Submit adverse event reports
- Renew product licenses periodically (every 5 years)
- Maintain GMP compliance
- Undergo audits, when required
- Notify TFDA of any changes such as formulation, packaging or TRP transfer
Core Documents Required for Regulatory Affairs Process In Taiwan
A structured dossier that meets both regulatory and localization standards must be submitted to register a product with the Taiwan Food and Drug Administration (TFDA). The following documents are typically required for product registration and may vary depending on the product category. Partnering with experts offering regulatory affairs consulting services can help ensure all documentation is accurate, complete, and aligned with TFDA expectations.
- Completed TFDA Application form with accurate product classification
- Product formula /composition with detailed ingredients and their percentage
- Manufacturing process and flowchart with GMP-aligned documentation explaining the product production, control and packaging process
- GMP certificate for drugs/health foods or Quality System Documents (QSD) for medical devices
- Free Sale Certificate (FSC)
- Product labels and packaging in traditional Chinese including the product details (such as name, dosage, ingredients, usage, warnings, manufacturer/importer information)
- Clinical efficacy data, if applicable (for new drugs, high risk devices or products with health claims)
- Power of Attorney to represent the applicant locally by appointing a Taiwan Registered Agent (TRP)
- Product samples or pictures reflecting the labelling and packaging clearly
Documents Preparation Tips
- Maintain a standard documentation structure to reuse for all the submissions.
- Ensure all the foreign documents are translated to Traditional Chinese.
- Use recognized international GMP or ISO certificates to avoid redundant inspections or reworks.
- Prepare eDossier according to the TFDA e-submission portal formatting and submission guidelines.
Why Artixio As Your Regulatory Affairs Consultant In Taiwan?





Artixio supports end-to-end product registration in Taiwan with local insights, regulatory accuracy, and full-cycle support—helping your products reach the market without delays or compliance risks. We offer expert regulatory affairs and compliance consulting services to guide you through every step. Connect with our Taiwan regulatory affairs consultant today for a smooth and timely approval process.
FAQs
1. Can the clinical data from other countries be reused for product registration in Taiwan ?
2. How long does it take to get product registration approval in Taiwan?
3. Can a product have multiple license holders in Taiwan?
4. Are there fast-track options for urgent or innovative products?
5. What happens if there are changes in manufacturing site or product composition post-approval?
Still Have Questions ?
Specialized Regulatory Affairs Services Across Multiple Industries
Expert Regulatory Services To Streamline Compliance

Regulatory Intelligence & Strategy

Medical & Technical Writing

Publishing & Submission






Regulatory Affairs Across Multiple Countries

India

Singapore

Mexico

Brazil

Vietnam

Malaysia

Argentina

Colombia

Taiwan

China

European

Thailand

Indonesia

Philippines

USA

Japan

Qatar

South Korea
Insights from Artixio - Tips & Articles

Cell and Gene Therapy
Cell and Gene Therapy Products are modern treatments to treat diseases. Treatment of these...

Cosmetics Regulations & Registration
Germany is one of the biggest cosmetics markets in Europe, but entering the market...

Why Medical Device Submissions
Introducing a new medical device to the international market requires that you not only...

