- Home
- >
- Services
- >
- Registration
Life Sciences Product Registration Services
We support life sciences companies with product registration and regulatory approvals across global markets. We assist with product registration and approval consulting services for life sciences industries across global markets. We offer tailored support from planning to post-approval compliance.
Product Registration & Approval Consultant Worldwide
We work with life sciences companies to bring products to market efficiently while meeting regulatory requirements. Our regulatory team combines hands-on expertise with smart tools to manage classification, documentation, and approval processes.
Whether it’s a drug, device, diagnostic, biologic, or cosmetic, we help you move through global regulatory systems with confidence. Every region has its own rules. We help you meet them without slowing you down.
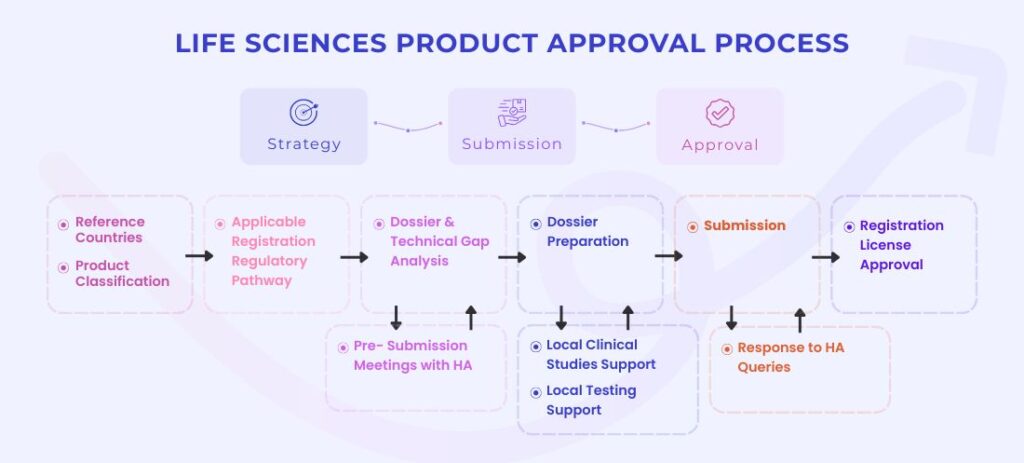
Life Sciences Product Approval Process
Strategy -> Submission -> Approval
Strategy -> Submission -> Approval
Tech-Assisted Regulatory Intelligence Driven Services Platform
QuriousRI is an AI-enabled regulatory intelligence platform that supports product registration strategies and activities across global markets.
Commitment to Excellence
With experience across multiple markets, product types, and therapeutic categories, Artixio applies proven regulatory practices to deliver efficient and reliable solutions for its customers.
Connecting Innovation with Global Markets
At Artixio, we help life sciences companies bring innovations to global markets efficiently. With deep knowledge of regional regulations and market dynamics, we simplify the regulatory process, so patients and end users can access the products they need, wherever they are.
Why Artixio For Life Sciences Product Registration?
- Successful Approvals and Product Lifecycle Maintenance for 5000+ products
- Intelligence, Insights & Experience Driven
- HA & Professional Network to Navigate Gray Areas
- Tech Assisted Approach
- Standardized, Checklist Based
- Efficient Project Management
- Milestone Based Approach
- Centralized Multi-Country / Multi-Product Strategy for Efficiency
FAQs
What are the key considerations for Product Registration in Global Markets?
In order to register new products (pharmaceuticals, devices, biologics, cosmetics, nutraceuticals, animal health etc), manufacturer need to take certain aspects in to consideration such as, applicable product classification, product registration regulatory pathway based on reference country, documentation requirements, local clinical studies / testing requirements, registration timelines and cost, local representation requirement, translation requirements, document notorization / legalization requirements, forms to be filled, GMP requirements, import license requirements, labelling and advertisement regulations among others.
What are the challenges faced during Product Registration in Global Markets?
International manufacturers face multiple challenges during product registration including, lack of knowledge of regional regulations, reliable legal representative with proven experience, subject matter expertise with Therapeutic Area / Product Category specific experience, transparency of information, Health authority experience, regional cultural and language nuances, on ground administrative support, accurate product classification, insights from health authorities related to unclear / ambiguous regulations, warehousing, distribution process clarity, import related challenges among others.
What are the requirements for Local Marketing Authorization Holder?
In most countries, foreign companies are required to appoint an authorized local representative that could be an individual or a company. In order to act as a Marketing Authorization Holder, the local company is required to hold certain licenses related to pharma business. Artixio, through its local offices and its affiliate network can support MAH services.
What are the additional local studies required for global Product Registration?
Depending on the product’s approval status in the country, reference country in scope, therapeutic area, complexity of the molecule, countries may require additional data. The data may include clinical studies data from local population, bioequivalence testing studies from local labs, medical devices testing and others. Artixio has a network of hospitals, clinical trial organizations, testing labs to support your local testing requirements, if applicable.
How will Artixio helps us with Product Registration in international markets?
Artixio has a centralized Regulatory Projects team with experience in managing multi-country registration for multiple products from diverse categories. The Projects team, together with our regional regulatory experts will work with you to understand your product, its current approval status against the requirements in countries of interest. They will help you through the registration process right from classification, gap analysis, HA meetings, dossier preparation and submission till approval.
Still Have Questions ?
Specialized Registration
Services Across Multiple Industries
Expert Regulatory Services To Streamline Compliance

Regulatory Intelligence & Strategy

Medical & Technical Writing

Publishing & Submission






Registration Across Multiple Countries

India

Singapore

Mexico

Brazil

Vietnam

Malaysia

Argentina

Colombia

Taiwan

China

European

Thailand

Indonesia

Philippines

USA

Japan

Qatar

South Korea
Insights from Artixio - Tips & Articles

Cosmetics Regulations & Registration
Failure to fulfil the regulatory requirements will lead the cosmetic product to be rejected,...

Cell and Gene Therapy
Cell and Gene Therapy Products are modern treatments to treat diseases. Treatment of these...

Cosmetics Regulations & Registration
Germany is one of the biggest cosmetics markets in Europe, but entering the market...

