- Home
- >
- Services
- >
- Pharmacovigilance
Local Qualified Professional for Pharmacovigilance (LQPPV) Services

Services We Offer
Local safety operations management as Deputy QPPV for global pharmaceutical and biotech companies
Pharmacovigilance in international markets is key to global pharmaceutical and biotech products manufacturers to ensure products remain safe for the patients as well as remain compliant with local regulations. Health authorities from countries globally mandate a local contact for pharmacovigilance, a qualified professional, to monitor, report safety concerns and submit periodic safety reports to the authorities. Further, the local QPPV person is also responsible for managing the health authorities communication and participating in safety audits, when required, working closely with the QPPV in the global PV team. Our LQPPV services include the following.
Artixio’s Local QP Pharmacovigilance Services:
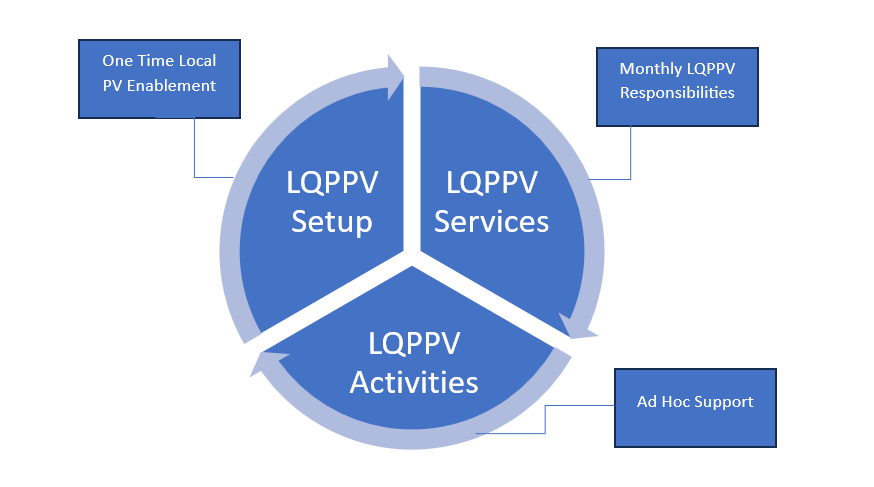
Local Qualified Person for Pharmacovigilance (Local QPPV) Setup
- Set-up of project objectives, budget, scope and completion of contract and SDEA
- Kick-off meetings
- Coordinate review and sign-off of LQPPV qualification and responsibilities
- Preparation of LQPPV & Deputy of LQPPV nomination documentation
- Nomination of LQPPV & Deputy of LQPPV to NCA as required by local requirements
- Set-up and access to all project-specific and general safety mailboxes as required
- Set-up of project-specific regulatory trackers
- Set-up of ICSR trackers
Local Qualified Person for Pharmacovigilance (Local QPPV) Services
- Acting as a local contact point for Local Health Authority and Qualified Person for Pharmacovigilance & Deputy of QPPV
- Handling Local Health Authority Requests and send safety notification (significant safety issues) to health authority
- Sending monthly reports and ongoing documentation/reconciliation of LQPPV activities
- Where required by local legislation this will include 24/7 availability, including back-up personnel
- Competent Authority Website Monitoring and PV related Regulatory Intelligence
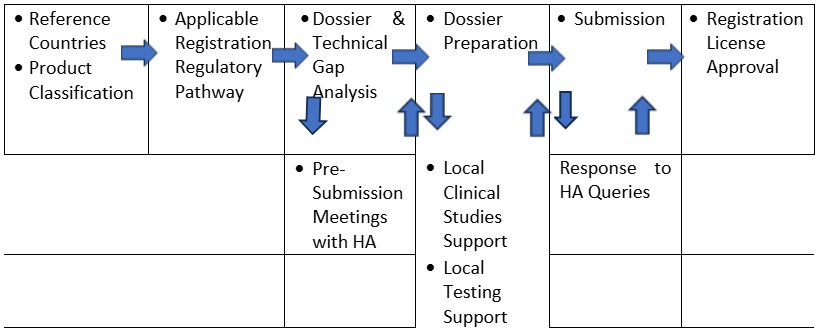
Strategy -> Submission -> Approval
Local QPPV Activities
- ADR Collection and reporting to, including but not limited to collection, translation of documents
- Handling of Local ICSRs (Receipt, tracking, forward to MAH/business partner)
- Requesting follow-up information from the reporter (ICSR follow-up)
- Handling of other enquiries (MI, PQC, etc.)
- Submission of documents to local Health Authority
- Translation of documents
- Attending Conference calls, training sessions and monthly meetings
- Participating in audits or inspections, CAPA management
- Conducting medical inquiry management
- Submitting and managing local PBRERs, PSMFs or RMPs and local RMPs/annex
- Training on global SOPs
- Tracking and delivery of pharmacovigilance training to local partners
- Any additional activities
- Setup, update, review, and implement SOP
- Setup, review and develop PV agreement
- Perform local/global literature screening
- Handle product Quality complaint with/out Adverse event
- Daily monitoring of safety mailbox
- Development, update and submission of PSMF
Seamlessly Augment Global Pharmacovigilance with through Artixio’s Local Pharmacovigilance Services through LQPPV
Our 100+ Pharmacovigilance experts based locally in 70+ key global markets to support ongoing safety monitoring and reporting to ensure product safety and compliance with local regulations. Coupled with AI enabled regulatory intelligence and monitoring to ensure both local and your global PV teams are up to date with latest local regulations for PV compliance.
Leverage Artixio’s Glocal Expertise with Seamless Global Delivery
- Successful Case Studies Local Qualified Professional for Pharmacovigilance Services supporting global clients
- Diverse Products Experience across Complex Products Categories and Therapeutic Areas
- Strong Regional Expertise in 70+ markets with 15-35 years of experience
- Health Authority Experience in Navigating through Complex, often Ambiguous Regulations
- Intelligence driven, global pharmacoviglance services supported by regulatory intelligence platform, QuriousRI
- ISO 9001:2015 quality standards governed services
- Customer focused, flexible services and solutions
- Flexible Business Model including project based, managed services and FTE staffing model
- Hybrid-Local-Centralized Support across Safety functions
Local Safety Compliance Delivered
Driven by key regional regulatory insights for safety, Artixio enables patient safety through proactive local pharmacovigilance strategies.
Centralized Regulatory Intelligence for Pharmacoviglance Regulations
Centralized, AI enabled platform for regulatory monitoring for updates related to pharmacovigilance from health authorities worldwide
Commitment to Excellence
With proven experience from successful management of pharmacovigilance processes and key learnings from different markets across product and therapeutic categories, Artixio is committed to excellence by implementing best management and pharmacovigilance practices geared towards efficient, reliable solutions for our customers’ pharmacovigilance needs.
FAQs
What are the key responsibilities of local qualified professional for pharmacovigilance (LQPPV)?
An LQPPV acts a local safety officer and deputy to QPPV from the global pharmacovigilance team to setup a local PV function in line with local regulations for safety. LQPPV ensures 24×7 availability to record any safety incidents and report to the global PV team. LQPPV acts as local contact person for the health authorities handling authority requests and sending safety notifications. In addition, the LQPPV supports many activities such as handling enquires, local ICSR, ADR collection and reporting, coordinating with local distributors and providing training on safety SOPs to local teams, submitting and managing local PBRERs, PSMFs, RMPs and other documents.
How do you support Local Pharmacovigilance across multiple countries?
Artixio is among leading providers of Regulatory and Safety services in emerging markets such as APAC and other regions. With over two decades of experience in the region and solid network of pharmacovigilance professionals, Artixio has built a platform to deliver on demand safety and regulatory services to global clients. We have in-house team of safety professionals and an extended list of affiliates based locally experienced in managing local safety for global leading brands. Our team capabilities coupled with AI enabled regulatory intelligence platform helps teams stay updated with latest regulations related to pharmacovigilance. We support both project based a staffing model through fractional or full time contract based local PV experts on monthly / annual basis.
What challenges do companies face in managing local pharmacovigilance and how do you address them?
- Finding right resources with experience in managing local PV
- Bilingual experts with PV experience with proficiency in both local language and English
- Lack of clarity of local PV regulations
- Lack of clarity of audit process from health authority of PV processes and systems
- Availability of fractional or part time retainer PV resources as most professionals are available for full time
- Lack of access to network and pool of professionals with vetted profile
- Lack of updates on latest local regulations and changes in regulations related to PV compliance
How do you stay updated with global regulatory changes related to pharmacovigilance?
Artixio has internal regulatory intelligence platform, QuriousRI, that tracks global regulatory changes related to pharmacovigilance. The platform hosts,
- Current regulations, directives, regulatory insights
- Track changes in regulations, new guidelines, notifications from health authorities worldwide related to pharmacovigilance, post market surveillance and products safety
- Insights by our Regional Regulatory Experts from their experience, attendance to Conference, Seminars, Trade Shows, HA Organized Trainings and Events
- Artixio’s expert opinions and Case Studies
- Key insights from Artixio’s experience, reports and responses to queries
Still Have Questions ?
Get expert answers tailored to your needs.
Specialized Registration
Services Across Multiple Industries
Expert Regulatory Services To Streamline Compliance

Regulatory Intelligence & Strategy

Medical & Technical Writing

Publishing & Submission

Product Registration

Investigation New Drug Application (IND)

New Drug Application (NDA)

Post Approval Lifecycle Maintenance

Artwork and Label Review

Ad Promo Review
Registration Across Multiple Countries

India

Singapore

Mexico

Brazil

Vietnam

Malaysia

Argentina

Colombia

Taiwan

China

European

Thailand

Indonesia

Philippines

USA

Japan

Qatar

South Korea
Insights from Artixio - Tips & Articles

From QSR to QMSR:
The medical device’s domain marked one of its biggest evolutions by release of transition...
April 1, 2026

Fragrance Ingredients: AI Strategies
Fragrance, though classified as cosmetics, follow a diverse compliance metrics governed by regulations from...
March 16, 2026

Probiotics – Strategies for
Globally probiotic products have shown exception growth driven by growing awareness of health benefits...
March 13, 2026

