The medical device’s domain marked one of its biggest evolutions by release of transition from Quality System Regulation (QSR) to Quality System Management System (QSMR). The main aim here was to harmonize US regulations with international standards.
This transition update was published on 31 January 2024, which entailed all the requirements and provided a transition period of almost 2 years for the manufactures to align with. And then on 2 February 2026, the transition came to an end, and an official update was published making it mandatory for the for the medical device manufacturers in the US to comply with the QSMR requirements right from their manufacturing phase to post-marketing surveillance.
Reading further will guide you in brief about the complete transition process of QSR to QSMR and the new requirements for the manufacturers to comply with.
What Was QSR?
The Quality System Regulation, also known as QSR, was introduced in 1996 and implemented in 1997. The QSR was introduced under 21 CFR in part 820, which entailed the quality system for medical devices in the US. It described the current Good Manufacturing Practice (cGMP) requirements, providing a brief overview of design, production and distribution for medical devices regulating in the US.
The implementation of QSR ensured the safety and quality of medical devices. Some of the key components of the QSR include:
- Production and process controls
- Corrective and preventive actions (CAPA)
- Documentation
- Compliant handling and post-marketing surveillance
- Design controls
Need For QSMR Transition:
The growing need to harmonize the global regulatory medical device standards along with the US regional standards, led to the transition of QSR to QSMR.
Many regulatory authorities worldwide were following the global standards for medical devices, but the US needed to comply with both the standards, regional as well as global, for its medical devices. This increased the overall regulatory burden and was becoming a very time-consuming method for compliance.
Thus, to overcome these regulatory challenges, the QSMR was brought into force, which introduced new global cGMP requirements through the ISO 13485:2016. The FDA thus modified the 21 CFR part 820, referencing it to ISO 13485:2016, to the new Quality Management System Regulation.
This transition mainly aimed to:
- Reduce the regulatory burden
- Achieve global harmonization
- Achieve timely compliance with devices
- Achieve easy market entry
- Enhance product safety and efficacy
- Build a quality-oriented device manufacturing process along with the product build quality.
Aligning with ISO 13485 for Global Consistency
The global harmonization of QSMR by the US FDA marked significant alignment with international quality standards of ISO 13485:2016. Thus, this international quality standard laid the foundation for QSMR. This led to the requirement of the incorporation of quality in production and distribution throughout the manufacturing process of medical devices.
This alignment helped the US manufacturers streamline compliance across multiple global regulatory bodies in Canada, Australia, the EU, the UK, etc.
Through this approach, the US FDA introduced the QMSR, aligning the 21 CFR part 820 with the ISO 13485:2016 standards and making this the regulatory foundation for medical devices regulating in the USA.
The transition is not simply a renaming of QSR to QMSR.
Instead, the FDA:
- Amended 21 CFR Part 820
- Incorporated ISO 13485:2016 by reference
- Retained certain FDA-specific provisions necessary under the FD&C Act
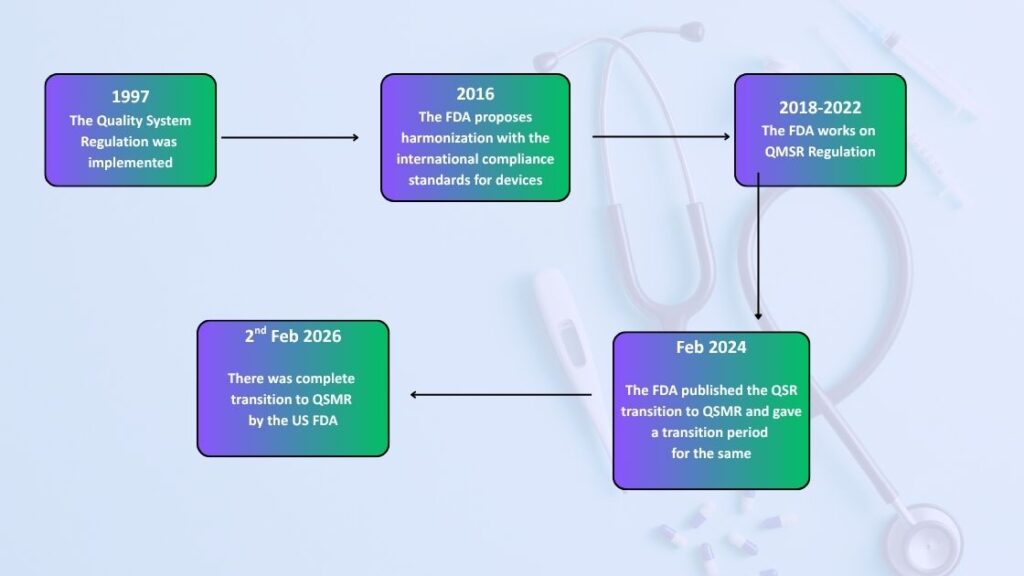
Timeline From QSR to QSMR:
The image above clearly explains the transition timeline of QSR to QSMR. Below are the steps of the transition timeline mentioned:
- In 1997, the QSR was published and implemented for medical devices regulating in the US.
- In 2016, the FDA aimed for global harmonization, hence a proposal for ISO global harmonization was proposed.
- In the period from 2018 to 2022, the FDA worked on the QSMR regulation.
- In the year 2024 Feb, finally the QSMR was published, and time was given for transition to the manufacturers.
- Then recently, on 2nd Feb 2026, the QSR was completely transitioned to QSMR and the amendments to 21 CFR part 820 was finalized with reference to ISO quality standards.
Key Changes from QSR To QMSR:
The transition from QSR to QMSR is not a complete overhaul change; it’s just some modifications in the 21 CFR part 820 aligning with the ISO 13485: 2016. Thus, it is essential for medical device manufacturers to completely understand the minute details and thus ensure timely compliance at each step of product lifecycle.
Below is the given table explaining the key differentiating features of QMSR from the outdated QSR:
| ASPECT | QSR | QMSR |
| Aim | Establish FDA specific quality requirements for medical devices governing in the US |
Harmonize the US quality requirements with the global regulatory quality standards
|
| Legal basis | 21 CFR part 820 |
Amended 21 CFR part 820 along with ISO 13485: 2016 incorporation
|
| Regulatory approach | FDA specific quality requirements |
ISO 13485 based quality requirements
|
| Global harmonization | Limitation alignment with global standards |
Complete harmonization with the international ISO standards
|
| Quality system structure | Detailed FDA based QSR sections |
Detailed QSMR requirements incorporating ISO clauses
|
| Documentation | Device specific documentation |
ISO pattern quality management documentation
|
| Terms | FDA specific terms such as DMR, DHR, DHF,etc | ISO oriented terms |
| Risk management | Risk mainly emphasized in design and final product |
Risk emphasized through the device lifecycle and quality incorporated at each step
|
| Inspections | FDA Quality System Inspection Technique (QSIT) |
FDA inspections according to new QSMR pattern and inspection for ISO certificate carried by independent accredited certification body
|
| Audits | Internal audits and management reviews largely protected from FDA inspection | FDA allowed to review audit data |
| Certification | There is no ISO certification requirement |
ISO certification is not mandatory, but the compliance criteria align with requirements of ISO certification
|
| Transition timeline | Implemented since 1997 |
Rule published in 2024 and effective from 2 Feb 2026
|
| Post market surveillance | Less integrated lifecycle view |
Lifecycle-based QMS with stronger post-market focus
|
Impact on Medical Devices Manufacturers:
This transition has affected the medical device manufacturers greatly, requiring them to align their manufacturing systems according to the new amendments in the 21 CFR part 820, aligning with the ISO 13485:2016.
The following are some of the impacted points on medical device manufacturers:
- The manufacturers need to review and update their quality management system in reference to the ISO clauses.
- The medical device manufacturers need to train their team to align their procedures with the QSMR.
- All the device documents and quality need to be prepared in reference to the ISO requirements.
- The system needs to be changed to a risk-based quality approach; it needs quality to be built throughout the product lifecycle.
- Suppliers and outsourcing device manufacturers should be more focused. They should undergo in-depth qualifications, monitoring, and evaluation.
- The manufacturer needs to note the changes in the documentation process of Device Master Records (DMR), Device History Records (DHR), and Design History Files (DHF), etc., according to the ISO clauses.
- The manufacturers need to prepare inspections according to the new amendment, as it has changed the inspection method and criteria as well.
Those device manufacturers who already possess an ISO certificate will find it easy to comply with the new requirements; those who do not have one will be required to perform an in-depth gap analysis to comply with the new requirements.
Future Of Medical Device Quality Systems:
The transition to QSMR marked a significant regulatory global harmonization for medical devices in the US. Through this transition, the FDA aimed to modernize the regulatory framework and make it easy for US devices to be regulated in any other country without any compliance issues.
Through this amendment in 21 CFR part 820, the US FDA emphasized the risk-based quality approach throughout the product lifecycle. This risk-based quality approach is expected to expand further in the future.
Further technologies may expand and advance further to monitor and achieve quality at each step of the product lifecycle. Risk analysis tools and techniques may advance in the near future. Automated processes may be developed to detect the quality of loopholes in the system. There may arise a unified system to reduce regulatory burden and achieve timely global compliance.
Thus, the future holds much more development in the field of medical devices, making devices the global emerging harmonized products.
FAQs:
1. Is ISO certificate required for QSMR compliance?
No, ISO certificate is not mandatory; the QSMR compliance criteria require quality compliances according to the ISO 13485.
2. Does QMSR involve the FDA site inspections?
The QSMR transition does not replace FDA inspections with ISO audits. Hence, the FDA inspections shall still take place.
3. According to the new QSMR transition, is there a need to rewrite all the previous documents?
No, not all documents need to be rewritten. There are some structural changes with the documents that need to be aligned according to the ISO clauses.
4. How does QMSR affect supplier management requirements?
The supplier management should meet the risk-based supplier evaluation and monitoring processes in reference to the ISO standards.
5. Does QSMR affect medical devices’ registration and approvals?
No, the QSMR only deals with the quality management system of the medical device lifecycle and has no link with the approval pathways for the same.