If you’re planning to bring a nutraceutical into South Korea, one thing becomes clear pretty quickly — the process depends a lot on how your product is classified and whether your ingredients are already recognized.
In Korea, these products fall under what’s called Health Functional Food (HFF), and the rules are handled by the MFDS. Some products move faster through approval, while others take longer, especially when new ingredients are involved.
In this article, we’ll walk through how the system works, what the registration process looks like, and a few areas where companies usually get stuck.
South Korea MFDS Regulatory Authority
In South Korea, dietary supplements are called Health Functional Food(HFF) and they are regulated by the Ministry of Food and Drug Safety (MFDS). The MFDS formulates policies for functional health foods and assesses their ingredient usage. It also sets labeling guidelines for health supplements and nutraceutical products.
Collaborating with the National Institute of Food and Drug Safety Evaluation (NIFDS), MFDS oversees the application and evaluation of functional ingredient use in dietary supplements. The Korea Health Supplement Association (KHSA), a public organization, supports both industry and regulatory entities in their endeavors.
Some of the key regulations of MFDS that are applicable for the Health Functional Food are given below:
- Health Functional Food Act
- Enforcement rule of Health Functional Food Act
- Enforcement Decree of the health Functional Food Act
- Regulation about Recognition of Functional Ingredient and of Standard and Specification for Health Functional Foods
- Regulation regarding the Functional Ingredient Approval for Health Functional Food
- Labelling Standard for Health Functional Food
- Health Functional Food Code
Contact us For
What Are Nutraceuticals As Per MFDS?
We know that nutraceuticals have different names like the food supplement, health supplements, dietary supplements… but in South Korea this is called Health Functional Food.
Health Functional Food are products which are derived from functional ingredients (a substance that provides health benefits) that are nutritious and beneficial to the human body.
Nutraceutical Product classification South Korea
Since Health Functional Foods are derived from functional ingredients, the functional ingredient classification given below:
- Ingredients notified in the Health Functional Food Code: Those that are registered in the Health Functional Food Code. It can be used by anyone if it satisfies the manufacturing, functionality and other related requirements.
- Ingredients that are recognized individually by the MFDS: Ingredients that are not registered within the Health Functional Food Code. The business operator must submit the safety as well as other relevant data and must be approved by MFDS. Only companies that have approval can use this ingredient.
Requirements For An Ingredient To Be A Functional Ingredient
An ingredient is said to be a functional ingredient if it is:
i) Processed raw material where the origin is from microorganism, plant, animal, or water.
ii) Extract of or purified substance of any of the ingredients mentioned above (i)
iii) Synthetic duplicate of purified substance of any of the above-mentioned ingredients (ii)
iv) Combination of any ingredients mentioned in the above points.
v) Functional ingredients should be specified under the standards and specification of functional ingredients of the Health Functional Food Code
vi) The functional ingredient must be recognized as per Article 15 of Health Functional Food Act and Regulation on Approval of Functional Ingredients on Health Functional Food.
The Dietary reference intake for Koreans can be found in the Annex, Table 3 of Health Functional Food Code.
Clinical Trial Of Food Supplements, MFDS
MFDS require scientific evidence for Health Functional Food for functional ingredients that are not publicly notified or any modifications that concern the safety and effectiveness of the ingredient and for new ingredients that are not recognized.
Food Supplement Registration In South Korea
Health Functional Food (HFF) Registration pathway involves,
- HFF with ingredients registered with MFDS.
- Safety and Efficacy Assessment.
- Submit an application for HFF Registration.
- Registration of manufacturing site.
- Receive HFF approval.
- Post-Market Surveillance.
Documents required for nutraceutical product registration
- Application Form.
- Nutraceutical Product Information.
- Ingredients in Nutraceutical Information.
- Nutraceutical Formulation Details.
- Manufacturing and Quality Control.
- HFF Safety Data.
- HFF Efficacy Data.
- Nutraceutical product Labelling and Packaging.
- Product Samples
- Authorization Letter and Power of Attorney.
Timeline and Fees associated in registration of nutraceutical in South Korea
The timeline and the fee are given below:
| ITEM | TIMELINE | FEES |
| Registration of business License | Review period 3 working days. | 28,000KRW |
| Registration of new raw material | New-120 days Change-60 days | New-1,900,000KRW Change-800,000 KRW |
| Registration of manufacturing | Review period 3 working days | No fees |
** Kindly note that the fee is subjected to change. It is advised to confirm the current fee by visiting MFDS official website. **
Manufacture of Health Functional Food, MFDS
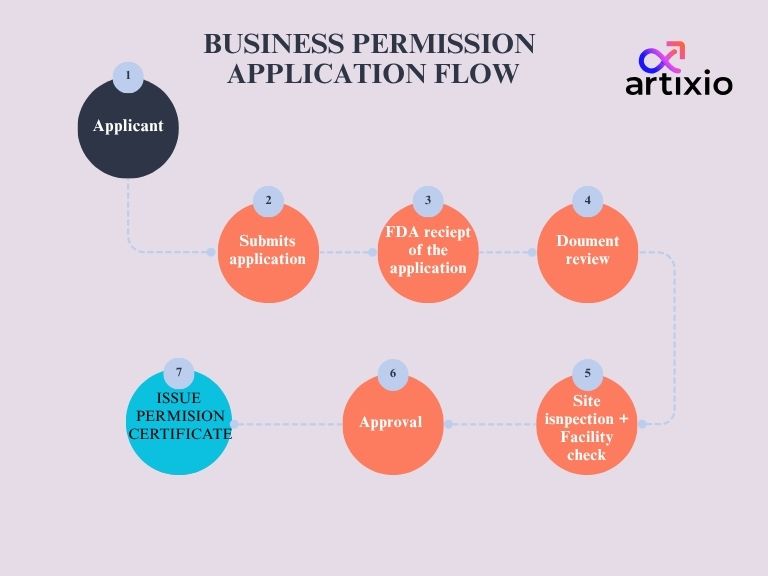
Any business person who intends to manufacture and/or sell the health functional foods must obtain permission from MFDS for each business place. To obtain permission, the business person must submit a business permission application (Form 1). In addition to this, the documents must also be submitted to the Commissioner of the Food and Drug Administration.
After review of the documents the Commissioner of Food and Drug Administration will issue a business permission certificate in Form 2. In case, the business permission certificate is worn out or is lost, then the business person must submit a business permission certificate reissue application in Form 4. The flow of business permission application is shown in the image below.
Documents for Health Functional Food – specializing business
The following are the documents to be submitted for Health Functional Food – specializing business.
- The product type and the manufacturing method document
- List of main apparatus & Machinery along with the arrangement plan of the manufacturing facilities
- Land use plan confirmation and business administrative register certified copy
- Quality management appointment report as per Article 16 of the Enforcement Rule of Health Functional Food Act.
- A certificate of education completion as per Article 13 (2) of the Act, when applicable
- Water analysis written result
Documents for Health Functional Food – venture business
The following are the documents to be submitted for Health Functional Food – venture business.
- Copy of venture business confirmation
- Technical data regarding the functional ingredient and the health functional food components.
- The product type and the manufacturing method document
- Quality management appointment report as per Article 16 of the Enforcement Rule of Health Functional Food Act.
- A certificate of education completion as per Article 13 (2) of the Act, when applicable
- A document which is on the manufacturing entrustment agreement with health functional food specializing business.
Import of Health Functional Food
Anyone who intends to file an import report as per the provisions of Article 8(1) of the Act, (which states that anyone who wishes to import Functional Health Foods, must file an import declaration) must submit a Health Functional Food Import Report in Form 19.
Appropriate documents must be submitted to the Regional Food and Drugs Administration Commissioner or to the Head of the National Quarantine Services. 5 days prior to the expected date of arrival of the Health Functional Food the import report can be filed. After receiving an import report for the health functional food, they examine the health functional food.
This is done with the examination methods stated in the Enforcement Rule for Health Functional Food Act. In Form 20 is the health functional food import report certificate issued if the product is compliance satisfied.
What happens if it is non-compliant? Let us take a look at the below image that explains this.

Documents Required For Import Report
The documents required are as follows:
- A copy of the Certificate of Examination or written result of the examination
- A packaging paper which bears a Korean label or the documents stating the Korean label contents can be submitted if the prior is difficult due to the transportation packaging etc..
- As per Article 11 (1) 7 of the Enforcement Rule of the Food Sanitation Act, an identity preserved handling certificate or the manufacturing country government recognized certificate to have an equivalent effect to that of the prior certificate. This is limited in cases where the food, subject to genetically modified labelling, does not have any genetically modified food label.
Some Important Applications And Timeline & Fee Associated:
Here is the list of some important applications concerning the Health Functional Food:
| Application | Treatment Period | Fee |
| Business permission | 14 days | 50,000 won |
| Business Permission Certificate Re-issue | Immediate | 5300 won |
| Modified matters on Health Functional Food Manufacturing Business |
|
30,000 won |
| Business Report | Immediate | 28,000 won |
| Business Report certificate Reissue | Immediate | 5300 won |
| Business Discontinuation Report | Immediate | Free |
| Health Functional Food Import Report | Approx. 20 days | 20,000 won |
Ingredients Subjected To Review
Manufacturers can use publicly notified functional ingredients for its health functional foods products provided that it must comply with the Standards and Specifications stated in the code. As per the Article 15 (2) of the Health Functional Food Act, certain ingredients are subjected to review to grant recognition of functional ingredients. They are as follows:
- Ingredients that are not notified to the public in the Standards and Specification for Health Functional Foods
- Any health claim addition to the functional ingredient that is notified to public in the Standards and Specifications for Health Functional Foods.
- Any alterations in the intake of such notified functional ingredients or to the manufacturing standards of the functional ingredient.
- Any additions, alteration or matters that are related to the safety or effectiveness of the functional ingredients.
MFDS Labelling Requirements For Health Functional Foods
According to The labelling Standard for Health Functional Foods, MFDS, the labelling requirements are as follows:
- The letters “Health Functional Food” or figure that stands for “Health Functional Food”
- Name of the product
- Business establishment’s name and place
- Sell-by-date
- Storage method
- Net contents
- Nutritional information including the nutrient, content and percent of recommended daily allowances
- Function information including the functional component and its content
- Raw material name and content.
- Consumption method and amount
- Any warning notice
- A statement indicating that it is not a medicine used for disease prevention or cure.
A detailed version is provided in the Labelling Standards for Health Functional Food regulation in the MFDS Official Website.
MFDS Administrative Fines For Health Functional Foods
An administrative fine not exceeding three million won will be imposed on any person who is falling under certain conditions. Some of the conditions are as follows:
| Condition | In accordance with |
| Fails to report a file on modification of permitted matters | Article 5(2) of the Act |
| Fails to report the modification of matters | Under Article 6 (2) |
| Fails to comply with the matters that is to be observed by the business entities |
Under Article 10 (1) 1 and 5 & who violates Article 10(2)
|
| Interrupt with the duties of quality control manager | Under Article 12 (3) |
| Failure in reporting the appointment or dismissal of quality control manager | Article 12 (4) |
| Fails to undergo training | Under Article 13(1) through 3 |
Post-marketing Activities Nutraceuticals in South Korea
The main activity or the main concern after the launch of a food supplement or any other product into the market is the adverse events or side effects that it begins to show in the real-world setting. A Health Functional Food Side Effect Reporting System was established by the MFDS to manage the side effects of the health functional food. It collects incidences of side effects and other related information.
MFDS conduct strong follow up management via the collection, inspection and monitoring of the food supplements that are distributed. Moreover, it is important for the manufacturers to conduct self-quality checks to ensure that they are in compliance with all the relevant regulations and requirements.
The Food Traceability System has been implemented by the MFDS to analyse, track and recall when hazards occur to food supplement products. It also aids in providing more accurate information to the consumers. This is becoming mandatory by phase for businesses that import or manufacture health functional food products with annual sales exceeding a certain limit.
Apart from this GMP compliance, labelling compliance and all other relevant requirement compliance must be made sure even after product entry into the market. Regulatory warnings, penalties, fines or even withdrawal from the market are the results of non-compliance.
Conclusion
Unlike other products, the food supplements are products that are meant to benefit the human body. For instance, vitamins and minerals when consumed within the limit only bring benefits to the body. This is why people rely on nutraceuticals. But it is important to find the non-adulterated products. This is why the MFDS has made certain regulations and require complete compliance. This ensures the safe and quality entry of food supplements into the Korean Market and consumers.
Artixio is here to help you with the food supplement registration in South Korea. Our regional experts and professional network always take you a step ahead with the MFDS regulations. We provide assistance right from development to market entry and maintenance. Connect with us through info@artixio.com for an expedited registration process.
FAQ’s
Q. What is the role of the Health Functional Foods Deliberation Committee?
A. The role of Health Functional Foods Deliberation Committee is to examine, deliberate and advise the MFDS regarding the following matters: policies, standards and specifications, labelling and advertising and other important matters regarding the health functional foods.
Q. Do MFDS offer any monetary rewards? If yes, at what condition and how much?
Not only the MFDS but also the Mayor of the Special Self-Governing City or the Governor of the Special Self-Governing Province, or the head of a Si/Gun/Gu may pay a monetary reward. It is awarded to a person who accuses or reports a person who violates Article 5 (1), 6 (2), 23, or 24 to the relevant administrative agencies or investigation agencies. The reward is such that it is not exceeding ten million won.
Q. What is the approval criteria of a functional ingredient for a Health Functional Food?
A. The 2 approval criteria are i) The ingredient must be in compliance with the Health Functional Food Act and ii) Safety and functionality must be made sure and proven scientifically.
Q. What is the time period for receiving response from MFDS when applying for a permission?
A. The MFDS must notify the applicant within 20 days of permission or non-permission, acceptance or nonacceptance after receiving an application for permission under Article 5 paragraph (1) of the Health Functional Foods Act. The time period is reduced to 7 days for report of modification as prescribed in the Article 5 paragraph (2) of the Act.
Also Read:




