With the increasing health consciousness among the Brazilian population of 215 million, there has been increasing demand for nutraceuticals and food supplements which marks the growth of the Brazilian supplements market. Brazil serves as a rich source of biodiversity, thus allowing the Brazilian supplements to use the natural ingredients of the Brazilian biodiversity.
This also serves as another factor in enhancing the demand for Brazilian food supplements globally. These supplements and nutraceuticals in Brazil are regulated by ANVISA (Agência Nacional de Vigilância Sanitária), ensuring the safety and quality of the nutraceuticals before they reach the consumers.
This article thus aims to brief you about the detailed requirements of ANVISA regulations for dietary supplements and nutraceuticals in Brazil.
Overview Of ANVISA in Brazil:
- Nutraceutical regulations in Brazil are regulated by the ANVISA which is Agência Nacional de Vigilância Sanitária also known as the National Health Surveillance Agency. ANVISA is responsible for confirming the safety and quality standards of food supplements.
- The ANVISA defines food supplements as “Products for oral administration, presented under pharmaceutical dosage forms intended to supplement the diet of healthy individuals with nutrients, bioactive substances, enzymes or probiotics, alone or in combination.
- The ANVISA sets guidelines for dietary supplement registration in Brazil, including all the parameters of production, distribution, and marketing.
- In 2018, the ANVISA introduced some new guidelines for the harmonization of the Brazilian food supplement regulations.
- For more details regarding ANVISA, visit the ANVISA official website.
BRAZIL Health Authority and Legal Representation
Nutraceutical and dietary supplements regulations in Brazil falls under the purview of the National Health Surveillance Agency (Agência Nacional de Vigilância Sanitária – ANVISA). ANVISA is responsible for overseeing and enforcing health regulations related to food, medicines, and other health-related products, including dietary supplements.
Brazilian Supplements Classification By ANVISA:
The aim of ANVISA is to categorize nutraceuticals as one of the subcategories of food, distinguishing it from medicines. According to RDC No. 243/2018, these food supplements are included under “Suplemento Alimentar” which means Food Supplement. Thus, the following are the nutritional categories of food supplements:
- Vitamins and Minerals
- Amino acids, proteins
- Probiotics
- Enzymes
- Plant extracts
- Other approved ingredients
The food supplements may comprise one of the above category nutrients or maybe a combination of two to three nutrients.
ANVISA Nutraceutical & Dietary Supplements Registration Process In Brazil
Pre-registration
Before initiating the formal registration process with ANVISA, manufacturers or importers of dietary supplements should gather essential documentation and information about the product. This includes details about the ingredients, product composition, safety data, and evidence supporting any health claims (non-therapeutic claims).
Steps For Supplement Registration In Brazil:
Document Requirements in Brazil by ANVISA For Nutraceutical & Dietary Supplements Registration :
| Module | Document Requirements |
| Module I – Administrative Documents | Company registration and legal representation documentation
Technical Responsibility documentation Proof of payment of applicable fees |
| Module II – Product Technical Documents | Product composition details
Manufacturing process description Quality control documentation Brazil (GMP compliance) Stability study data Packaging and labeling details |
| Module III – Product Safety and Efficacy Documents | Toxicological and safety data
Clinical studies (if applicable) Supporting evidence for non-therapeutic claims |
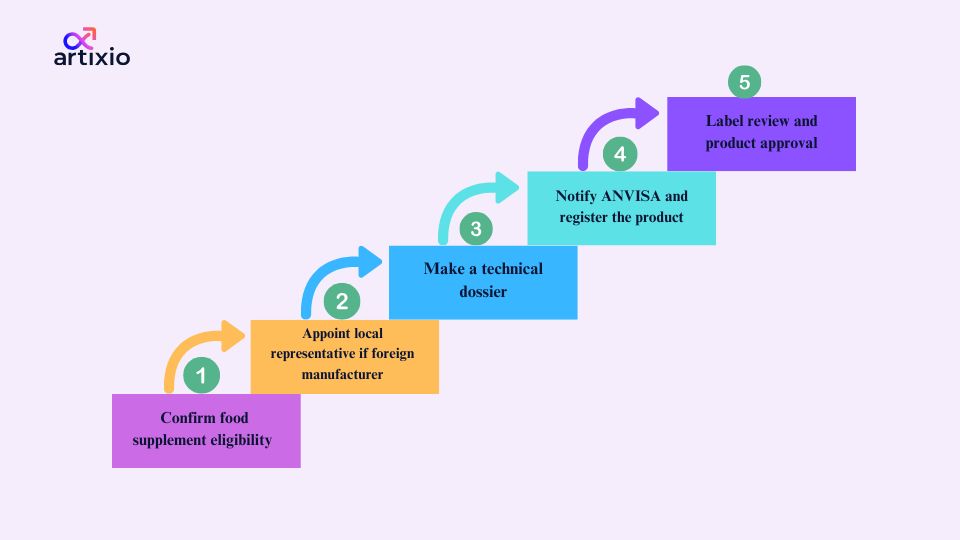
The steps involved in the ANVISA supplement registration process in Brazil are:
- Firstly, the food supplement should fulfill the “Suplemento Alimentar” requirement, that is the ingredients used should not have any therapeutic properties and they should be listed in the ANVISA-approved list.
- If the food supplement is manufactured by a foreigner, he should appoint a local Brazilian representative to handle all the regulatory procedures.
- Then the technical dossier should be prepared that consists of all the details of the product including its safety data, manufacturing process, etc.
- After this, the manufacturer should notify ANVISA through its online portal about the marketing of the food supplement, if the product consists of a novel ingredient, it will require pre-market approval.
- Then the product’s label should be prepared and confirmed with its compliance with the ANVISA regulations.
- And then the product can finally be marketed.
ANVISA Supplements Labeling Requirements in Brazil
The labeling of food supplements shall have the following information:
I. The recommendation for use according to population group and age range in the case of children.
II. The quantity and frequency of consumption for each of the population groups indicated on the label.
III. Highlight and bold warning:
- “This product is not a medicine”
- “Do not exceed the daily recommendation of consumption indicated on the packaging”
- “Keep out of the reach of children”
IV. the instructions for storage, including after opening the package.
Dietary Supplements Import and distribution in Brazil
Once a nutraceutical or dietary supplement is approved for sale in Brazil, importers and distributors must also comply with Brazil’s health authority guidelines. They should have an operating license from ANVISA, follow good storage practices, and ensure proper transportation conditions to maintain product integrity.
Supplements Timeline and Associated Fees by ANVISA
The timeline for the registration process is 12-18 months and the fee associated with registration of dietary supplements in BRAZIL ranges from $100 USD-$ 2060 USD.
Dietary Supplements Post-Market Surveillance in Brazil
After the food supplement has entered the market, it is very crucial for the manufacturer to keep monitoring the effect of the product on the consumers by conducting post-marketing surveillance (PMS). Any adverse reaction, quality issues, or changes from the manufacturer like changes in the label, packaging changes, etc. should be reported to ANVISA.
Regulatory Non-Compliance Consequences in Brazil:
It is mandatory for food supplements to comply with the ANVISA regulations in Brazil. Violence of any regulatory requirements may lead to penalties from ANVISA. Some of the consequences of non-compliance involve:
- Product suspension: The product may be suspended from the Brazilian market.
- Product recall: The product may be recalled from the Brazilian market by the ANVISA to check for various parameters such as safety, quality, label, etc.
- Fines/Penalties: The violence of the ANVISA law may impose fines and penalties depending on the severity of the violation. The charges range from BRL 2000 to BRL 1.5 million.
ANVISA Supplement Guidelines 2025:
- As of the recent year, ANVISA regulates according to the regulation RDC No. 243/2018 with updates in labeling transparency, ingredient control, and product safety.
- Supplements are classified as “Suplemento Alimentar”
- The ingredients should be used from the approved list under the regulation IN No. 28/2018 and IN No. 29/2018.
- No therapeutic claims should be made.
- Nutraceuticals should strictly comply with the labeling, nutrient limit, and serving guidelines.
Regulatory Requirements For Supplements In Brazil:
Some of the key points of regulatory requirements for food supplements in Brazil are as follows:
- To confirm the food category of the supplement that is, it should come under the “Suplemento Alimentar” category.
- The nutraceutical should contain ingredients mentioned in the ANVISA-approved ingredient list, or if using novel ingredients it should be pre-approved before manufacturing.
- If a foreign manufacturer, he should appoint a local representative in Brazil for his regulatory works.
- The dossier should consist of all the information regarding the product’s safety, efficacy, quality, manufacturing details, etc.
- Before the market entry, the product should be notified via ANVISA’s SGAS system.
- The manufacturing should be conducted in a GMP-compliant environment.
- The label should be prepared in compliance with ANVISA guidelines.
- Post-marketing surveillance should be conducted and reported to ANVISA.
Conclusion:
Artixio is committed to assisting companies in achieving regulatory compliance and market success in the Brazil nutraceutical and dietary supplements industry. Contact us today to learn more about how Artixio can support your regulatory needs in the Brazil. Contact us at info@artixio.com today!
FAQs
How much time is required for the food supplement to get notified by ANVISA?
The ANVISA takes a few weeks to get notified with the food supplement notification, and if the nutraceutical has a novel ingredient, it requires 3-6 months for pre-marketing evaluation to get approved.
Are herbal food supplements allowed in Brazil?
Yes, certain plant-origin supplements are allowed in Brazil, according to which are listed in the ANVISA allowed ingredient list.
Does ANVISA allow food supplements for children in Brazil?
Yes, but food supplements for children involve more strict and severe regulations such as pediatric approved, age-specific supplements, ingredients according to the allowed concentration, etc.
How can the food supplement notification status be tracked in Brazil?
The notification status for food supplements can be tracked using Sistema de Gestão de Autorização de Suplementos (SGAS), which is the online portal of ANVISA to monitor the notification updates.