- Home
- >
- All Countries
- >
- Taiwan
Life Science Compliance Services in Taiwan
Taiwan regulatory compliance services for life sciences firms—practical guidance from submission to successful approval. Turn your concept into commercialization with our end-to-end streamlined regulatory services.
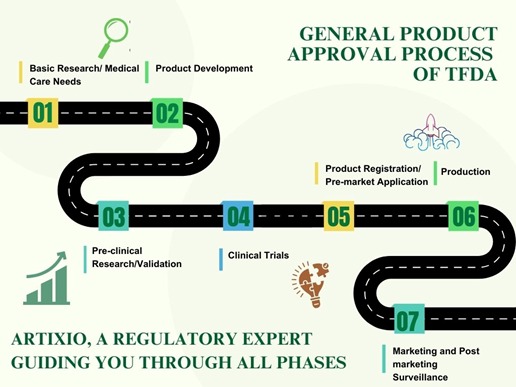
Your Partner for End-to-End Regulatory Support in Taiwan
We support companies across the life sciences sector with practical regulatory solutions tailored to every stage of the product journey. From development to registration and market entry, Artixio helps simplify the process and reduce delays. If you’re looking to fast-track approvals without cutting corners, we’re here to help.
Regulatory Authorities in Taiwan
You’ll find that in Taiwan, different health products are regulated by specific authorities. Most human health products—like medicines, medical devices, cosmetics, biologics, and food—fall under the Taiwan Food and Drug Administration (TFDA), which works under the Ministry of Health and Welfare. If you’re dealing with veterinary products, those are looked after by the Animal Health Inspection Division under the Ministry of Agriculture.
TFDA wasn’t always a single body. It was formed in 2010 by bringing together four separate units that used to handle pharmaceutical affairs, food safety, drug analysis, and controlled substances. By combining these, the government aimed to streamline oversight and make sure that both human and animal health products meet safety and quality expectations before reaching people or animals.
How We Help Meet Compliance Standards in Taiwan
Artixio is an ISO 9001:2015 certified company that provides quality and reliable services to our clients. We have region-based experts with 15-35 years of experience in guiding our clients through Pharmaceutical, Medtech, Beauty and Wellness, Food and in Animal health & Welfare sectors. Even Though we are still expanding our network and services across the globe, we have a well-established network in Taiwan. Our local Subject Matter Experts have produced a 95% of success rate for all the projects with their well-versed regulatory knowledge.
Where does our expertise come from?
Post-registration
Market entry services, Post market Surveillance, Product lifecycle management services, performance evaluation reports, Advertisement and promotional material review, Digital marketing and other ongoing support services.
Services through all phases
Registration
Pre-registration
Our Services



Why Artixio As Your Taiwan Regulatory Consultant?










FAQs
What are some common challenges faced by foreign manufacturers in Taiwan?
What is the RTF procedure?
What is the function of TFDA?
What are the criteria for an application to undergo priority review under TFDA?
For an application to undergo priority review process, it must meet any 2 of the following criteria: i) It must be new chemical entity, combination, indication or must have a new administration route, ii) Must meet the unmet medical needs (in the treatment of serious conditions) and should have a significant clinical advance if approved, iii) Addresses an unmet medical need and is under the special national scientific research and development programs.
What is PIC/S and is TFDA a member of PIC/S?
Industries We Serve in Taiwan
Our Global Reach: Serving Life
Sciences Clients Worldwide

India

Singapore

Mexico

Brazil

Vietnam

Malaysia

Argentina

Colombia

Taiwan

China

European

Thailand

Indonesia

Philippines

USA

Japan

Qatar

South Korea
Insights from Artixio - Tips & Articles

Cosmetics Regulations & Registration
Failure to fulfil the regulatory requirements will lead the cosmetic product to be rejected,...

Cell and Gene Therapy
Cell and Gene Therapy Products are modern treatments to treat diseases. Treatment of these...

Cosmetics Regulations & Registration
Germany is one of the biggest cosmetics markets in Europe, but entering the market...







