Cosmetic Regulatory Compliance Software for Global Markets
QuriousRI regulatory intelligence platform supporting cosmetic compliance workflows including ingredient safety checks, labeling, regulatory mapping, and multi-country compliance monitoring across global markets.

Services We Offer
Cosmetic Compliance Check Tool for Global Markets
Globally, cosmetics are regulated by health authorities to ensure that the products are safe to use by target population. Health authorities use regulations, cosmetic ingredients databases, chemical databases such as pubchem, expert opinions and research publications to ascertain that ingredients used in cosmetic product formulation are not prohibited, are safe to use under recommended daily limits.
Regulatory Affairs teams within cosmetic companies are enables for business to expand in domestic and international markets while ensuring the products stay compliant with regulations. Nevertheless, each country follows its own process, guidelines and references to evaluate the safety of cosmetic formulations, making it a time consuming, daunting task. Check your formulations more efficiently with our cosmetic ingredient compliance checker designed for global regulatory requirements.
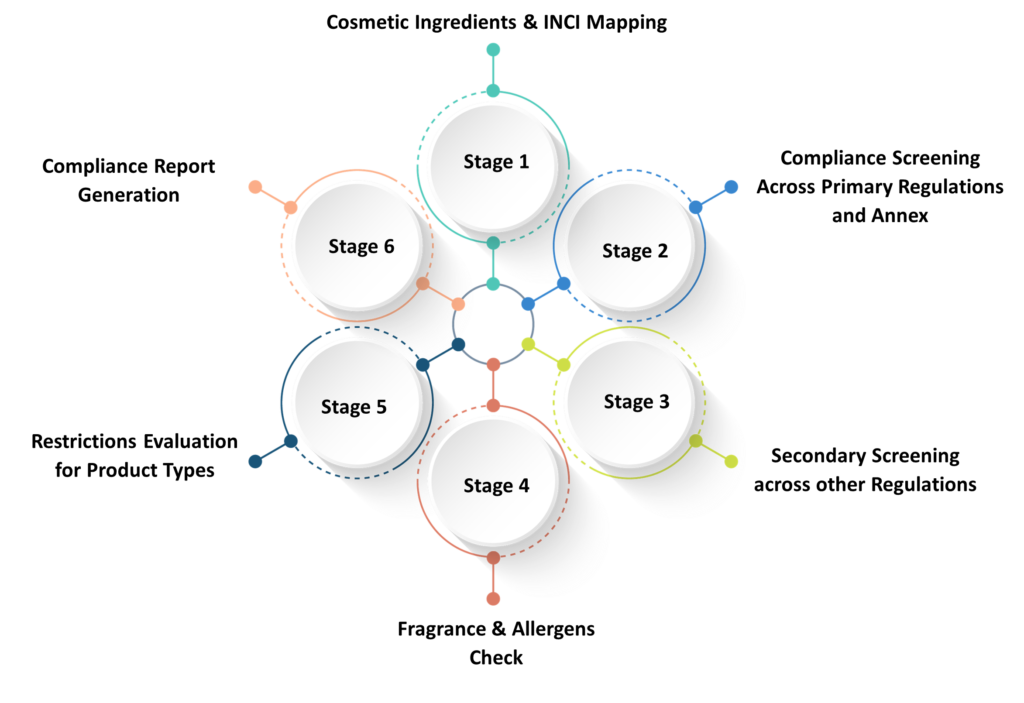
QuriousRI’s compliance check platform is an assistive technology that accelerates global compliance check for cosmetic products using six steps.
Key Features of our Cosmetic Compliance Software
Ingredient Compliance Checker
Global Regulatory Database (US FDA, EU CPNP, ASEAN, etc.)
Labeling Compliance Automation
Product Registration Management
Document & Dossier Management
Regulatory Alerts & Updates
Accelerate your ingredients compliance review and INCI mapping with a structured approach to ingredient validation across global regulations.
Why Cosmetic Compliance Software Matters For your business?
- Keep track of cosmetic regulations across different countries often requires structured systems to stay organized
- Managing regulations in multiple markets becomes easier when supported by a centralized software solution
- Important updates can be easy to miss when everything is tracked manually
- Delays in compliance checks can slow down product launches
- It reduces manual effort in checking ingredients and regulations
- It helps teams stay updated with the latest regulatory changes
- It improves speed and accuracy of product compliance checks
- It supports faster and safer product launches in global markets
How QuriousRI Fits Into Your Cosmetic Regulatory Workflow
- During formulation: Teams can check cosmetic ingredients against global regulatory databases and validate compliance early in the product development stage
- Before product launch: Formulations can be reviewed for market-specific requirements, helping ensure compliance before entering different countries
- During registration: Regulatory teams can refer to structured data and documentation to support submissions across markets
- After product launch: Ongoing monitoring helps track regulatory updates that may impact existing products
- Across teams: Everyone works with the same set of information, so there’s less back-and-forth and fewer mismatches between teams
- Replacing manual work: Instead of jumping between different documents and databases, teams can review everything in one place
QuriousRI brings these references together so you can review your formulation with more clarity and reduced back-and-forth iterations. Our cosmetic ingredient review platform is meant for teams that periodically checks ingredients against different regulatory lists.
Global Cosmetic Compliance Coverage Using our Tool
- US (FDA / MoCRA)
- EU (CPNP, Annexes, REACH)
- UK (SCPN / UK Cosmetics Regulation)
- APAC (ASEAN Cosmetics Directive, Indonesia BPOM, Malaysia NPRA, Singapore HSA)
- LATAM (Brazil ANVISA, Mexico COFEPRIS)
Continuous Update of Global Cosmetic Regulations and Ingredients Database
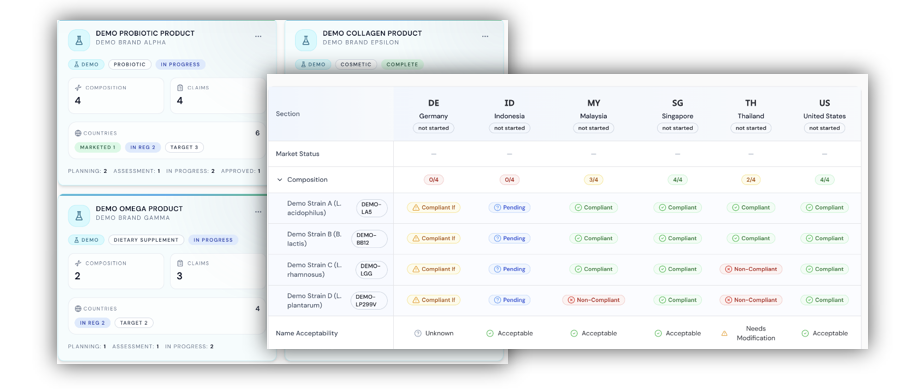
QuriousRI updates global changes in regulations for cosmetic products as well as ingredients, chemical and fragrance databases such as Cosing, pubchem, IFRA, REACH, Annexes, 21 CFR and other country specific guidelines globally across key markets. With a seamless automated workflow, regulatory teams can perform compliance evaluation or markets of their interest within the platform backed by references from health authorities and official sources.
Streamlining monitoring and risk impact assessment of cosmetic regulations across countries —QuriousRI, our cosmetic regulatory compliance software helps integrated the workflow under one platform.
Why Cosmetic Compliance Feels Complicated
Ingredients evaluation methods change depending on target markets, countries and health authorities. What works in one market may need a closer look in another, and the references are not available at one place; they are scattered across sources and often not available or available in local language.
Therefore, the review process ends up taking considerably significant time.
Who To Use Our Cosmetic Compliance Regulatory Intelligence Tool
- Cosmetic brands expanding globally
- Manufacturers & private label companies
- Regulatory affairs teams
- Importers/exporters
- Consultants managing multi-country compliance
How QuriousRI Simplifies Compliance Checks
QuriousRI helps you evaluate cosmetic ingredient compliance in a structured, faster way:
- Check ingredients against global regulatory databases
- Identify restricted or prohibited substances instantly
- Access references from official health authorities
- Evaluate compliance across multiple markets in one place
Built for Cosmetics Product Regulatory Teams
QuriousRI is used to:
- Review ingredients against multiple regulatory references
- Keep track of updates without manual monitoring
- Work across teams without scattered files
It’s designed to support the way regulatory teams already work, just with fewer gaps.
Go Beyond Compliance Checks
Once your formulation is evaluated, QuriousRI helps you:
- Monitor regulatory updates impacting your products portfolio
- Track compliance status across countries with global dashboard
- Get support from Artixio’s 300+ global regulatory experts
Accelerate Your Product Launch
- Reduce manual effort.
- Improve accuracy.
- Expand into new markets faster.
Accelerate Your Product Launch
Reduce manual effort. Improve accuracy. Expand into new markets faster.
Leverage Artixio’s Global Expertise
- AI-Assisted, cosmetic ingredients compliance check for key global markets
- Hybrid, Regulatory Professional + Tech Model
- 300+ Regional Regulatory Experts for Expert Support
- Accelerate Time to Market
- Expand to global markets seamlessly
- Executive dashboard for global cosmetic compliance status
- ISO 27001 certified and GDPR compliant
- Dedicated deployment for enhanced data security
Real Time Compliance Monitoring for Cosmetic Products
Once you have assessed the ingredients / formulation compliance, monitor your products’ compliance against changing regulations and updates ingredients safety limits, publications or findings to ensure your products stay compliant in global markets.
FAQs
What are the key features of the QuriousRI Compliance Check tool?
What are the challenges faced by regulatory professionals during cosmetic regulatory compliance review and product registration?
How can I evaluate the number of countries where I can register or market my cosmetic product?
Still Have Questions ?
Specialized Registration
Services Across Multiple Industries
Expert Regulatory Services To Streamline Compliance

Regulatory Intelligence & Strategy

Medical & Technical Writing

Publishing & Submission






Registration Across Multiple Countries

India

Singapore

Mexico

Brazil

Vietnam

Malaysia

Argentina

Colombia

Taiwan

China

European

Thailand

Indonesia

Philippines

USA

Japan

Qatar

South Korea
Insights from Artixio - Tips & Articles

Cosmetics Regulations & Registration
When a product is classified as a medicine or a borderline product, it becomes...

Cosmetics Regulations & Registration
Cosmetic notification in the Philippines is frequently delayed because the labels are non-compliant with...

Cosmetics Regulations & Registration
The Vietnam cosmetic launch may be postponed due to minor problems. This may be...