Advanced medicines, such as cell, gene, and tissue-engineered therapies, are revolutionizing modern medicine. In Singapore, Cell, Tissue, or Gene Therapy Products (CTGTP) are regulated by the Health Science Authority (HSA). Based on the risk category, CTGTP is classified as class-1 CTGTP and class-2 CTGTP.
The class-2 CTGTP is a high-risk category and requires product registration with the HSA. In January 2026, HSA updated the guidance on the registration and variation process for class-2 CTGTP. The guidance emphasizes procedures for manufacturers/applicants to verify compliance with updated requirements before marketing products in Singapore.
Registration Process for Class 2 CTGTP in Singapore
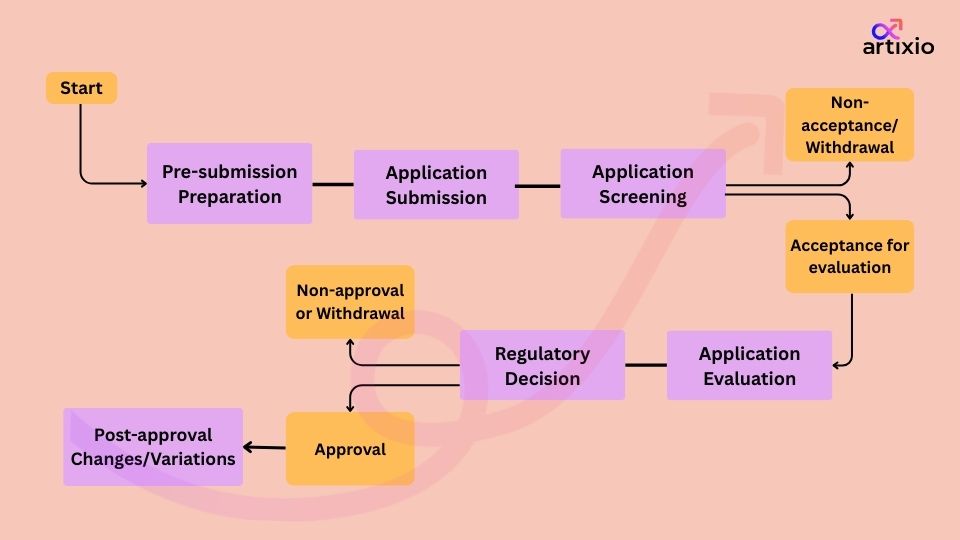
As class 2 CTGTP requires registration, it is important to comprehend the registration procedure before submitting an application to HSA. The following flowchart describes the complete flow of the registration process.
Application Types
For the application submission, there are three application types for Class 2 CTGTP.
| Application Types | Conditions |
| New Drug Application (NDA)-1 |
The first strength of the CTGTP which is currently not registered in Singapore.
|
| NDA-2 |
1. For the first strength of a product containing: New combination of registered CTGTP New dosage forms, such as suspension for intravenous infusion and powder for injection New presentation, such as single-dose vials, freezing bags, and pre-filled syringes New formulation New route of administration 2. For products that do not fall under the requirements for NDA-1 or NDA-3. |
| NDA-3 |
For subsequent strengths of a CTGTP that has been registered or submitted as an NDA-1 or NDA-2.
|
Evaluation Routes
Assessment of the application is evaluated via a specific evaluation route. Based on the evaluation route document requirements, evaluation time, and fees are determined.
| Evaluation Route | Conditions |
| Full evaluation |
New CTGTP, which has not been approved by any HSA’s comparable overseas regulators. Pre-submission notification is required for this route |
| Abridged evaluation |
New CTGTP that has been approved by at least one of HSA’s comparable overseas regulators.
|
Pre-submission Notification
Applicant is required to submit a pre-submission notification at least two months before the full evaluation application via email at HSA_CTGTP@hsa.gov.sg. with the following information-
- Product name
- Active ingredients
- Summaries of Quality, Non-clinical and Clinical data
- Planned submissions in other countries
- Planned date of submission to HSA
Fee payment and Turnaround time
Applicants are advised to make payment through the GIRO (apply eGIRO).
Turnaround time (TAT) is the time required for the screening/evaluation process of an application, from the date it is submitted. It excludes the applicant’s stop-clock, the time taken by the applicant for responding to the additional information request.
Applicant can refer to applicable fees, TAT – CTGTP fees, and turnaround time.
Application Submission
Application dossier
Applicant must submit an application dossier with all technical documents, either in ICH-CTD or ACTD format, through SHARE.
The dossier format used in the registration of CTGTP will determine the format for the variation applications of registered CTGTP.
HSA provided the application checklist, which should be completed by the applicant-
Pre-market consultation
Applicants can book a consultation with HSA to seek feedback on the appropriateness of documents and product-specific regulatory requirements for registration of CTGTP.
Each consultation is restricted to one CTGTP and lasts for one hour duration. Request for the consultation booking must be submitted at least six months prior, and related documents need to be submitted at least 30 working days before the scheduled consultation date.
Screening and evaluation process
-
Screening of the application
During the screening stage, HSA verifies that the application type is accurate and the dossier is complete. After the successful screening of the application, this will enter the evaluation phase.
-
Evaluation phase
Once the dossier is accepted for the evaluation, HSA will issue a notice to the applicant, which marks the starting date for the evaluation timeline.
If HSA requires additional data or clarification, HSA will send an input request with a clock stop time of 20 working days. The applicant should respond with an appropriate answer to the query.
Variation applications
Based on the risk of the proposed change and the approval requirement, variation applications are classified into three types:
- Major variation 1 (MAV-1) application
- Minor variation 1 (MIV-1) application
- Minor variation 2 (MIV-2) application
The variation application must be submitted with the applicable evaluation route via SHARE. Applicants should look at the CTGTP fees and turnaround time.
-
Major variation 1 (MAV-1) application
MAV-1 applications cover the significant change related to the quality, safety, and efficacy of registered class 2 CTGTP and require prior approval. For examples:
Changes in dosing regimen
Changes in the patient group
Changes in approved indications
Applicants can submit up to three concurrent MAV-1 applications to each registered CTGTP at a time. The application must be completed as per the updated checklist with the relevant documents:
-
Minor variation 1 (MIV-1) application
This includes minor variations that are specified in part A of the MIV Checklist for Class 2 CTGTP, which requires approval before implementation.
-
Minor variation 2 (MIV-2) application
MIV-2 applications are further classified as:
MIV-2 (Notification) – This includes minor variations that are specified in part B of the MIV Checklist for Class 2 CTGTP, which are required notification before implementation.
MIV-2 (Do and Tell) – Specified in part C of the MIV Checklist Class 2 CTGTP, and no prior approval is needed; it can be implemented directly. Variations should be submitted within six months of implementation.
Conclusion
The updated guidance from the HSA streamlines the regulatory framework for the CTGTP registration and the variation process. As the complex, high-risk nature of Class-2 CTGTP requires proactive and strategic planning for full-filing evolving regulatory requirements throughout the product life cycle.
For applicants/manufacturers, it is necessary to select the appropriate application type, evaluation route, and classifications of post-approval changes in a pre-defined category to maintain compliance.
Artixio provides regulatory services in Singapore for dossier preparation, market approval, and life-cycle management of advanced therapy products, supporting faster and more efficient approvals. Contact our regulatory team in Singapore today!