The Health Sciences Authority (HSA) functions as the regulatory authority responsible for overseeing medical devices within the country, ensuring their safety, effectiveness, and quality. For businesses engaged in the manufacturing, importing, or wholesale distribution of medical devices in Singapore, the initial step in device registration involves obtaining a dealer’s license. Prior to issuing the dealer’s license, HSA will evaluate your compliance with Good Distribution Practice requirements.
Irrespective of the classification of device risk, every medical device must obtain a Dealer’s License from HSA. An exception is granted for specific low-risk Class A devices, which do not necessitate evaluation or approval through the standard process. Instead, an exemption form called the Class A Exemption List form must be filled out as part of the dealer’s license application.
Types of Medical Device Dealers in Singapore
Licensing of dealers is based on the activity performed by that company in relation to medical devices. There are 3 types of dealer’s licenses for dealing in medical devices.
- Manufacturer’s License
- Importer’s License
- Wholesaler’s License
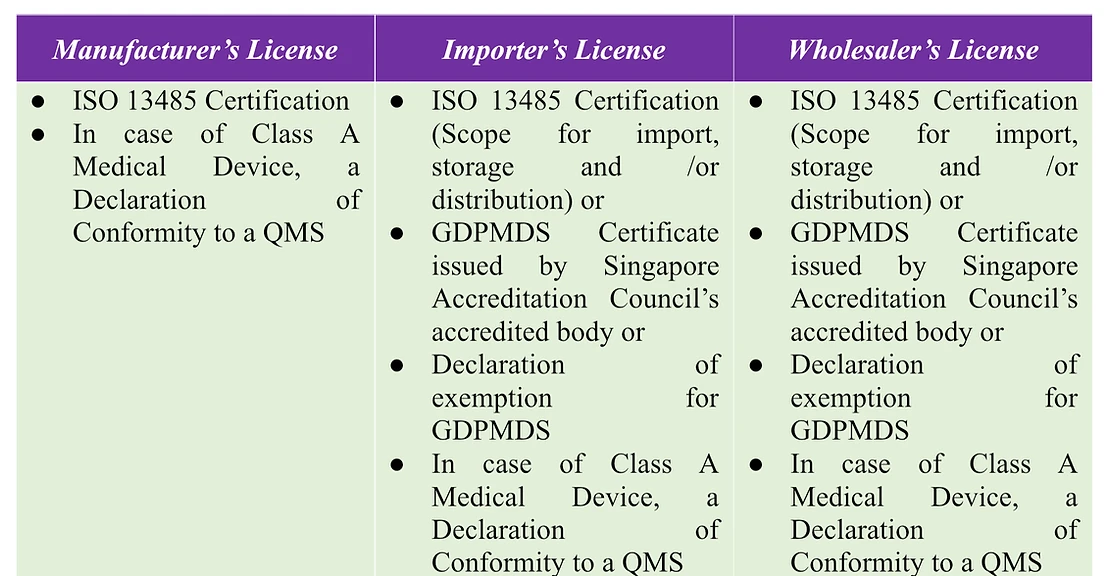
Manufacturer’s license
A Singaporean medical device manufacturer must obtain manufacturer’s license to create, package, label, and distribute medical devices. Once a manufacturer license is issued, they do not need an additional wholesaler’s license to distribute devices they’ve produced under their manufacturer’s license.
Importer’s license
A company importing medical devices into Singapore needs an importer’s license, which permits them to bring medical devices into the country. However, licensed importers must also obtain a wholesaler’s license if they intend to supply these devices on a wholesale basis.
Wholesaler’s Licence:
A company engaged in wholesale distribution of medical devices in Singapore, including export, must obtain a wholesaler’s license. This license enables them to supply medical devices wholesale, provide them for re-supply, or distribute them as commercial samples. Notably, a wholesaler’s license is unnecessary for supplying medical devices directly to end-users, such as in a retail store.
HSA Documentation Requirements for Obtaining a Dealer’s License for Medical Devices
To secure a dealer’s license for medical devices, businesses must provide essential administrative details and showcase the implementation of required Quality Management Systems. Companies with an Import/Wholesale license are not obligated to obtain a manufacturing license, as long as they comply with GDPMDS/ISO 13485 standards.
HSA Process for Dealer License Application
Here is the simplified summary of the process, facilitating businesses’ compliance with regulatory requirements for obtaining the Dealer’s License.
- The foreign manufacturers/importers must establish a legal subsidiary entity or appoint a local importer/wholesaler, or designate an independent third party.
- All dealers must be registered with the Singapore Accounting and Corporate Regulatory Authority (ACRA).
- To access the MEDICS e-service in MEDICS, applicants must obtain Personal Access Authentication, which enables them to log in via CorpPass (Singapore Corporate Access) or HSA PIN (HSA Personal Identification Number). CorpPass serves as a corporate digital identity for entities to interact with government online services, while HSA PIN is a password for overseas individuals provided by HSA.
- Companies seeking access to the MEDICS e-service system must also establish a CRIS account with HSA, granting them entry to the MEDICS portal for electronic transactions. The CRIS Company Account for MEDICS (Client Registration & Identification Services) is designed for local companies to access MEDICS.
- The applicant is required to complete the application and provide the following details:
- Applicant Details – Including name, passport number or NRIC (National Registration Identity Card) number, and contact particulars.
- License Particulars – Covering the dealer type (manufacturer, importer, wholesaler), device classification (Class A, Class A IVD, Class B/C/D, Class B/C/D IVD), site address, the certifications to be included, and the extent of operations. In instances where the applicant fulfills multiple roles, a separate application is necessary for each dealer category.
- Company Information – Encompassing the company name, address, Unique Entity Number (UEN), and the contact person’s information.
- Class A Exemption List – Relevant solely to manufacturers and importers, excluding wholesalers. The dealer is obliged to submit the declaration form. In the case of Sterile Class A devices, the dealer must confirm compliance with sterilization processes as per applicable international standards.
- Supporting Documentation.
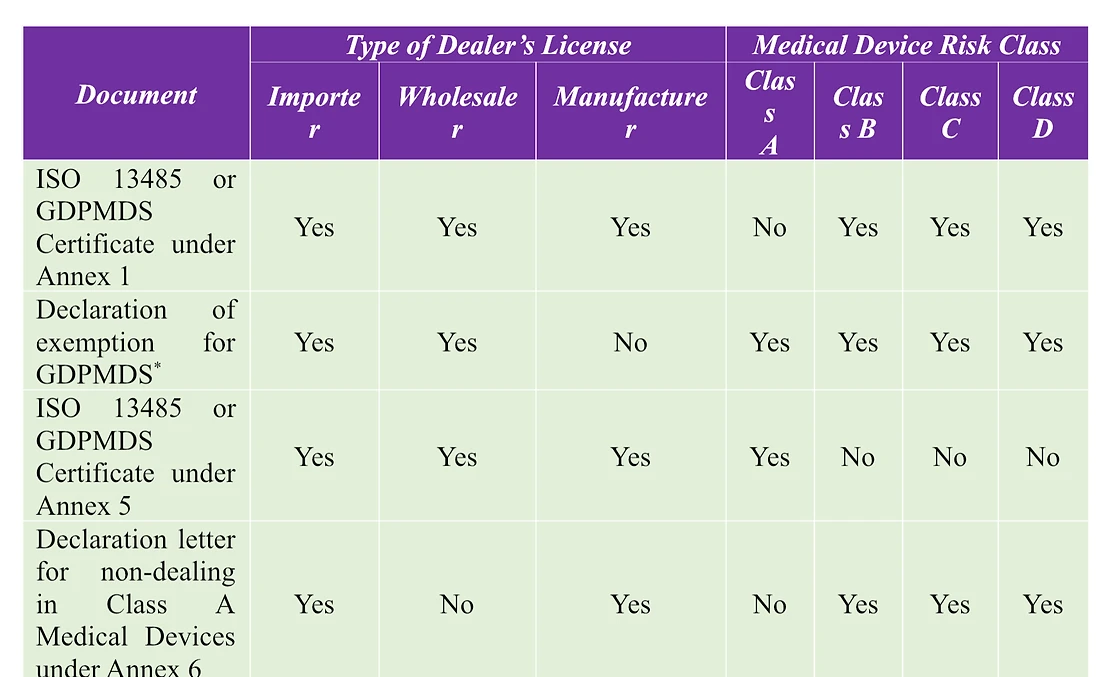
*only for medical devices for non-clinical or import for re-export purpose
The application includes a space for adding comments to the Licensing Control Branch (LCB) in case any dealers wish to provide extra information. The applicant is required to make a payment of $1,060. The payment can be made through eNETS or GIRO payment modes. HSA reviews the application and issues the appropriate Dealer’s License within a period of 10 business days. It’s important to note that this 10-business-day timeframe does not account for the time taken by the applicant to respond to any queries raised by HSA.
Modifications to Dealer’s License After Approval
Dealers have the flexibility to make alterations to their approved license. These changes can pertain to any information originally submitted as part of the licensing process. However, specific alterations, such as a change in Unique Entity Number (UEN), necessitate a fresh application. In such instances, a new CRIS account must be established. All licenses associated with the previous UEN will no longer be valid, and the applicant must initiate a license cancellation process.
When there are modifications to applicant information that affect other certificates and licenses, the details can be simultaneously updated in all relevant documents. For changes in company details, the necessary updates should be made in the “Change in Business Information” section of MEDICS.
Other changes, such as updates in device risk classification, Quality Management System (QMS) certifications, and declarations (e.g., changes in certification bodies, scope of certification, or QMS certificate renewals), require an amendment to the license application.
Any alterations in site information or the list of Class A medical devices being handled must also be kept current. If Class A medical devices are newly introduced to the product portfolio, the “Declaration Letter for Non-Dealing in Class A Medical Devices under Annex 6,” submitted during the original application, must be updated accordingly. The fee for HSA’s processing of these amendments is $160.
Renewing Your Dealer’s License by HSA
HSA issues the dealer’s license certificate, which is initially valid for one year from the approval date. During the original licensing process, applicants have the option to select either automatic renewal or manual renewal. If a dealer fails to renew the license in a timely manner, the license will be canceled, necessitating the submission of a new application. The renewal fee remains the same as the initial fee, which is $1,060. In case an entity holds multiple licenses, each one must be renewed separately.
Automatic renewal of licenses is exclusively available through the GIRO payment method. To ensure that the first GIRO deduction is processed in time for your license renewal, it is advisable to apply for GIRO at least 60 days before your license’s expiry. GIRO deductions are conducted 30 days before the license expires. If your GIRO application is not submitted in time for this deduction, you should initiate a manual renewal to ensure that your license is renewed promptly. It’s essential to maintain a sufficient balance in your GIRO account at least 45 days before the license’s expiration date to avoid unsuccessful renewal due to insufficient funds.
For those without GIRO, manual renewal is the alternative:
- You will receive two email notifications regarding the renewal before your license expires.
- You can manually renew your license in the MEDICS system starting 60 days before, and no later than 10 days from, the expiration date.
- Complete the renewal payment.
Securing a Dealer’s License for medical devices in Singapore is a critical step for businesses operating in this industry. The stringent regulations set by the Health Sciences Authority (HSA) guarantee the safety and quality of medical devices, necessitating strict adherence to compliance standards and meticulous document submission aligned with Quality Management System (QMS) requirements. Our experts are available to guide you through this process, providing detailed insights into the complex requirements and ensuring a smooth entry into the market.




