Working with CDSCO is part of almost every pharmaceutical registrations in India. The rules are well defined, but in reality, the process can still feel a bit scattered when you’re dealing with applications, approvals, and timelines.
This blog goes over how CDSCO pharmaceutical regulations in India actually work, especially from a registration point of view, and what companies should be prepared for during the process.
Classification of Drugs on the basis of Pharmacological Effect
In pharmaceutical effects they are classified on the basis of Effect they produce on the Human Body for ex- Analgesics (Relieves body pain), Antibiotics (Cure Infections), Antiseptics (Bacteriocidal or Bacteriostatic effect).
Legal Classification of Drug
- Schedule I – High potential of misuse or Addiction with no medical use
- Schedule II – High Potential of misuse but have some physical or psychological use
- Schedule III – Moderate to low potential misuse with psychological use
- Schedule IV – Low potential for dependence with good physical or psychological use
- Schedule V – The drugs used as analgesics, antitussive or antidiarrheal purposes with high physical use.
- Therapeutic classification in India is in Alphabetical order which includes Acidifiers, Analgesics, Chelating Agents, Gastrointestinal Agents, Minerals, Pharmaceutic Aid, Stimulants, Vitamins, Wax Emulsifiers and many more drug Classification.
- According to the types of medicine drugs are classified as Ayurveda, Unani, Yoga, Neuropathy, Homeopathy
- Pharmacology classification by Chemical Makeup includes Alcohol, Opioids, Benzodiazepines, Cannabinoids, Barbiturates
- Pharmacology Classification on the basis of Effect includes Depressent, Stimulants, Hallucinogens and Inhalants.
CDSCO governs the rules and regulations under the guidance of D&C Act and taken care by DGCI. In India, The manufacturer or importer needs to apply for CDSCO form 44 application along with required amount of fees as guided in Schedule Y of D&C Rules for manufacturing or importing any new drug or its formulation for the purpose of marketing or performing any clinical trial in India.

Looking for Pharma Regulatory Affairs Services in India? Our team can guide you through every step.
Functions Of CDSCO For Pharmaceuticals
The following are the function of CDSCO:
- New drugs and clinical trial approval.
- Registration, Import and licensing
- Banning drugs and cosmetics that serves as a threat for public safety.
- Amendments to the D&C Act and Rules
- Testing of new drugs
- License approving of r-DNA products, blood banks, vaccines, LVPs, and some medical devices.
- Grant of NOCs for export, test license and personal license
- Oversight and market surveillance
CDSCO SUGAM Portal
The online portal used for licensing or registration processes by CDSCO named as “SUGAM” for the ease of submission, registration, processing, filing applications and fast servicing through online platform. Submissions, reviews and permission or NOC Granting are the services provided by SUGAM portal.
Importer, Indian agent, Enterprise holders or corporate people can register and be an applicant on the SUGAM portal. Manufacturing unit can not directly register through the portal. The Responsible person of the organization after registering gets the approval/ Verification through E-mail. For registering the RP have to submit ID proof, Address proof documents to CDSCO office. There is no need to register again if the person has CDSCO Approval.
Understand the CDSCO SUGAM For Regulatory Submissions in detail.
Types of drug license under CDSCO Pharmaceutical Regulations in India
1. Wholesale drug license
2. Retail Sale Drug License
3. Manufacturing Drug License
4. Loan License
5. Import License
Documents required for obtaining drug sale license in India
a) Firm/Entity constitution, Company-Articles of Association (AOA), Memorandum of Association (MOA); LLP/Partnership- LLP or Partnership deed agreement
b) Owner/Proprietor/Director/Partner ID Proof
c) Property Ownership or Rental Agreement document
d) Any proof for storage, Refrigerator or Cold Storage availability.
e) Registered pharmacist affidavit
f) Premises Site plan and Key plan
g) Board resolution permit proof and challan copy
h) MPD 2021 Compliance Affidavit if the premise is located on DDA region
Documents required for retail sale pharmacist
a) Local pharmacy council registration
b) Letter of appointment
c) Qualification proof
Documents required for wholesale pharmacist
a) Letter of Appointment
b) Qualification proof
c) Certificate of Experience
Pharmaceutical Product Registration In India
Here is the CDSCO registration process for pharmaceutical products in India:
1. New Drug Approval
The Efficacy and Safety of the product is the first priority for approval process. It needs to check that the drug falls under the pharmacological activity and safe for human use. The CDSCO and Officer leader established Drugs Controller General (India) (DGCI).
Schedule Y under D & C rules give the information about requirements and guidelines for clinical trials which were further categorized as CATEGORY A (trials conducted in other market) and CATEGORY B (remaining ones fall under this category).
The drug approval process is comprised of two steps – a) Application for conducting clinical trial and b) Marketing Authorization request for drug from regulatory authorities.
The categories that adhere for the application process are Category A, B, C, D and E.
2. Certificate of Import Registration
CDSCO Certificate of Import Registration process is only for the importers wants to import drugs. Application is placed for issuing import registration certificate along with the required amount of fee to be paid with it. It can also be charged for the inspection fee and the DGCI either approve the GMP certification from the advanced countries such as EU, US or inspect on their own against Indian GMP. This process may take longer time.
3. License for Import
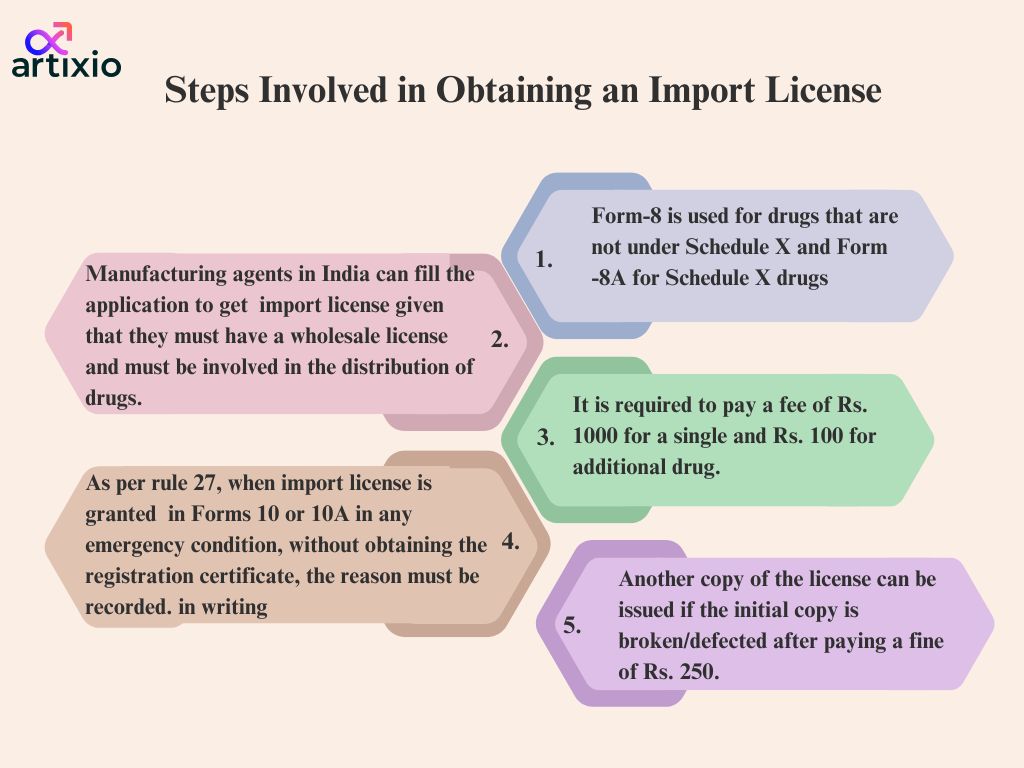
The import license is to be issued by the foreign importers specified in Rule 10 and Rule 10A. Rule 24 of D&C Rules deals with requirements for obtaining license. Application is placed for import license before placing the importer must have Wholesale Drug License and other required licenses according to D&C Rules. Generally within 3 Months from the date of application the license is issued.
If a drug has already been approved in places like the US or Europe, it can make the Indian review a bit smoother. CDSCO Authorities here may refer to decisions taken by agencies like the FDA or EMA while reviewing the product. It’s not a shortcut, but it does add weight to the application.
Clinical trial requirements in India
Clinical trials in India come under the scope of CDSCO. When a company plans to introduce a new drug, regulators usually expect some trial data generated within the country. This helps them check how the drug works for Indian patients before giving any approval.
Clinical Trial Approval: Before any study begins, the sponsor has to secure approval from CDSCO through the DCGI’s office. This is done by filing a Clinical Trial Application (CTA). The application includes the study protocol, the investigator’s brochure, ethics committee clearances, and other supporting documents needed to show that the design and safety measures are well thought out.
Ethics Committee Approval: Apart from regulatory approval, it is important to get approval from an independent Ethics Committee registered with CDSCO. This is essential to make sure that the trial will be conducted by protecting the patient safety, rights and well-being of the trial participants. It is the responsibility of the EC to ensure the trial is conducted in an ethical manner.
Compensation and Insurance: If any trial related injuries or deaths happen to the trial participants, an appropriate compensation must be provided. This is required to be done by the sponsor. A clinical trial insurance is compulsory to cover the liabilities (if any) that arise at the time of clinical trial.
Good Clinical Practice (GCP): It is a mandatory regulatory requirement to be in compliance with the GCP standards. Any clinical trials conducted must follow the GCP. This is the same for the clinical trials conducted in India as well. The principles of GCP are outlined in the International Council for Harmonisation (ICH-GCP). Compliance with GCP guarantees ethical and scientifically sound conduct of clinical trials and data reliability.
Informed Consent: Participants must be explained about the objectives, procedure, benefits and potential risk that can occur. No participants must be forced into a clinical trial. It must always be their own choice whether to be a part of the trail or not. So, informed consent must be obtained from each participant before their inclusion in the study. If the participant is not in a right state to give informed consent (children, unconscious state, diseases like Alzheimer’s), their legally authorized representative can provide the consent.
Safety Reporting: Prompt reporting of any adverse event that occurs during the trial should be reported to the Ethics Committee and the regulatory authority. This is required to be done by the Sponsor and investigator.
Clinical Trial Registry: Clinical Trial Registry – India (CTRI) is a publicly accessible clinical trial registry. Before the enrolment of the first trial participant, it is mandatory to register the clinical trials in CTRI as per the drug licensing authority, DCGI.
The permission to initiate the conduct of a clinical trial is granted in Form CT – 06 or Form CT 4A (automatic approval). This remains valid for a period of 2 years from the date of issue unless it is extended by the Central Licensing Authority.
Drug License & Manufacture Cost In India
There are different forms to be used for specific type of sale licensing. Application form 19 (19-A for restricted type of sale) is used to apply for license for wholesale and retail sale of drugs other than that specified in Schedule C&C (1) &X, Drugs specified in Schedule C&C (1) & not specified in Schedule X. For drugs under Schedule X, the application form in 19-C. Similarly, the fee structure also varies. The fee is as follows:
| Category | Type | Fees |
| Drugs: other than that, specified in Schedule C&C (1) &X | Wholesale
Retail Restricted |
Rs 1500
Rs 1500 Rs 500 |
| Drugs: Specified in Schedule C&C (1) ¬ specified in Schedule X | Wholesale
Retail Restricted |
Rs 1500
Rs 1500 Rs 500 |
| Drugs in Schedule X | Wholesale
Retail |
Rs 500
Rs 500 |
| Application for | Fees Applicable |
| Permission to manufacture new drugs, Finished formulation or API, for sale or distribution | Rs 5,00,000 |
| Permission to manufacture new drugs, finished formulation or API, which are already approved for sale or distribution | Rs 2,00,000 |
** The fees are subjected to change and must confirm it by referring to the official website before making any payments**

Timeline
The time required for Import registration certificate is generally 9 months from the date of application. It takes about 180 days for the approval or registration of new drug.
CDSCO Labelling Requirements For Drugs In India
The labelling requirements must be as per the Drugs and Cosmetics Rules 1945. Part IX specifies the requirements to be followed in detail. The manner of labelling is as follows:
- Name of the drug
- An accurate statement about the net content in terms of weight, volume, measure, number of units of contents, number of units of activity (as the case may be) and the weight, measure and volume be expressed in metric system.
- Content of active ingredients
- Manufacturer’s name and the address of the premises where the drug has been manufactured.
- A distinctive batch number, that is to say, the number of the particular batch from which the substance in the container is taken are recorded and are available for inspection. The figure representing the batch number must precede the words ”B. No.” or ”Batch No.” or ”Batch” or ”Lot” or ”Lot No.”.
- Requirements for labelling of Schedule X drugs, drugs for export, etc are mentioned under Part IX of the Drugs and Cosmetics Rules 1945.
How CDSCO Tracks Drug Safety Post Approval
Getting drug approval from CDSCO isn’t really the final step. Once pharma products are out in the market, that’s when post-approval tracking actually starts. In India, this comes under pharmacovigilance, where side effects or unexpected reactions get noticed during regular treatment and reported over time.
Not everything comes up during trials, so this stage matters more than it seems. If similar issues start showing up again and again, they usually get looked at more closely. Companies are also expected to keep sharing safety updates with CDSCO, especially in the early phase after approval, just to make sure nothing is missed.
Also Read: Phytopharmaceuticals Regulations In India
Conclusion
At Artixio, our team of experts try to understand every aspect of the novel or generic products in context of Indian market and provides high quality end-to-end solutions regulatory solutions for CDSCO registration process and post approval maintenance of pharmaceutical products.
Stay tuned to Artixio for more insights into pharmaceutical regulations and market trends, and reach out to us at info@artixio.com.
FAQ’s
Q. What is the fee structure for import registration of drugs?
1500 USD or Rs. 1,30,723 must be paid by the applicant as registration fee for manufacturing premises. 1000 USD or Rs. 87,148 is the registration fee to be paid for a single drug and for each additional drug for which the manufacturing site remains the same, an additional fee of 1000 USD must be paid.
Q. When is registration renewal or re-registration done?
Application must be submitted 9 months before the expiration date of the registration certificate for the renewal of the registration or re-registration.




