The Mexican population is seen getting very conscious about their health and early aging which has led to an increase in the demand for nutraceuticals in Mexico in recent years. Thus, nutraceuticals in Mexico serve as one of the major markets in Mexico which is expected to generate a revenue of $ 14.9 million by 2030 in the Mexican pharma market.
Mexican nutraceuticals and dietary supplements market is regulated by the COFEPRIS under the category of Foods and Non-Alcoholic Beverages.
COFEPRIS Nutraceutical Products Regulations
Mexico has special regulatory authorities to oversee the functioning of Mexican dietary supplement regulations. As nutraceuticals aim to promote healthy being, the Mexican government makes it mandatory to confirm the safety, efficacy, and quality of the nutraceutical products thus making it completely safe for the consumers. In Mexico, nutraceuticals are categorized under dietary supplements and enforced by health product laws which are regulated by government agencies.
The main authorities responsible for coordinating the efficient manufacture and distribution of nutraceuticals are as follows:
COFEPRIS Or Federal Commission For Protection against Sanitary Risks (Comisión Federal para la Protección contra Riesgos Sanitarios):
- COFEPRIS serves as the main authority for regulating nutraceuticals in Mexico.
- COFEPRIS must check and confirm that the nutraceuticals that are intended to be sold in the Mexican market comply with its guidelines so that a safe and quality-efficient product reaches consumers.
- COFEPRIS also investigates GMP compliance and clinical trial conduction.
SAGARPA (Secretariat of Agriculture, Livestock, Rural Development, Fisheries, and Food):
- SAGARPA is responsible for looking after the raw materials used in the manufacturing of nutraceuticals.
- It confirms that the raw materials used in nutraceutical manufacturing comply with the quality standards.
NOM (Official Mexican Standards):
- NOMs are a set of standardized regulations for the products and services to meet consumer safety.
- NOM-051 is made by the NOMs for the regulation of the nutraceutical labeling and health claims made on the label.
COFEPRIS defines Supplements as:
Products based on herbs, plant extract, traditional food, dehydrated or concentrated fruits, added or not, of vitamins or minerals, that can be presented in a pharmaceutical form and whose purpose of use is to increase the total dietary intake, supplement it or supply some components.
Supplements can contain- carbohydrates, plants, algae, dehydrated traditional food, metabolites, fatty acids, amino acids, and protein.
Supplements cannot contain- procaine, ephedrine, substance with pharmacological action, germanium, animal or human hormones, and yohimbine.
Planning to market Dietary supplements in Mexico? Access our COFEPRIS regulatory affairs services for end-to-end compliance support.
Pre-registration Requirement For Nutraceuticals in Mexico :
Before starting the registration process, companies must ensure that their products comply with all relevant regulations in Mexico and have the necessary documentation ready for submission.
COFEPRIS Nutraceuticals and Dietary Supplements Registration in Mexico
Follow the given steps below for the registration of nutraceuticals in Mexico:
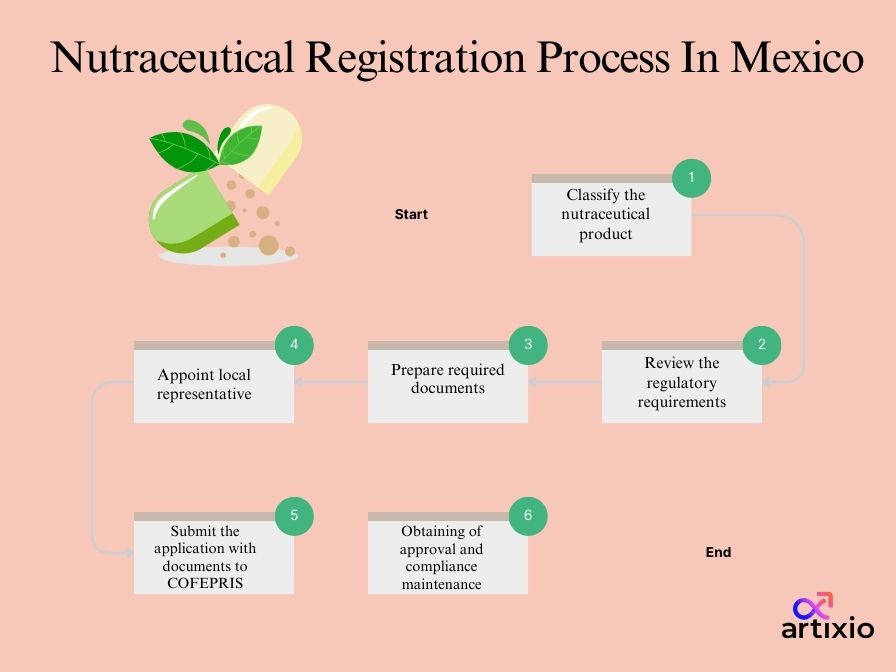
- Review the list of ingredients in the product.
- Review the label and develop with a client Mexico-compliant label.
- Classify the product.
- Prepare pre-consultation submission to COFEPRIS.
- Pre-consultation submission to COFEPRIS.
- Wait for approval/rejection/queries from COFEPRIS.
- Submission to COFEPRIS.
- If final approval – the product may be distributed and sold in Mexico.
Documents required by COFEPRIS For Nutraceuticals Registration:
- Product Information: Name, composition, intended use, and packaging details.
- Safety Data: Toxicological and clinical safety data.
- Quality Control: Specifications and analytical methods used to ensure product quality.
- GMP Compliance: Evidence of adherence to Good Manufacturing Practices.
- Labeling Information: Draft label artwork and compliance with labeling requirements.
Labeling Requirements for Nutraceuticals/Dietary Supplements in Mexico:
There are certain labelling requirements that nutraceuticals should follow, which are as follows:
The label should provide adequate and authentic information and must possess the following information on the label:
- Product’s name
- Ingredient’s list
- Ingredient quantities
- Its dosage
- Instructions for product use
- Scientific evidence
Approval in EU/US and Progressing Registration Mexico:
If a product is already approved in the European Union (EU) or the United States (US), it can expedite the registration process in Mexico. The submission of relevant documentation and approval from COFEPRIS.
Import and Distribution Requirements nutraceuticals in Mexico:
Only registered products are allowed for import and distribution in Mexico. Importers and distributors must comply with all Mexican regulations and maintain records of the products they handle. National product registration is lenient. Where only the production facilities must be notified.
Timeline and associated fees nutraceuticals in Mexico:
The registration timeline for nutraceuticals is associated with the completeness of the application. In general, it may take a period of several months to a year or sometimes even more. As per the fees associated, it might cost 215 USD.
Post-marketing Activities Of Nutraceuticals in Mexico:
Post-marketing activities in Mexico involve activities such as:
- Monitoring and reporting of adverse effects.
- Timely renewal of product registration.
- Conducting audits and inspections.
- Seeking approval if the product undergoes any changes in the manufacturing process, labeling, packaging, etc.
List Of Allowed and Prohibited Ingredients For Nutraceuticals In Mexico:
| Allowed Ingredients | Prohibited Ingredients |
| Vitamins and Minerals within their specified range. | Pharmaceutical ingredients |
| Botanical ingredients | Hormones |
| Amino extracts | Controlled substances |
| Fatty acids | Caffeine exceeding the limits. |
| Probiotics and Prebiotics | Unapproved ingredients |
| Fiber | Cannabis derivatives |
| Enzymes | Steroids |
Popular Nutraceuticals Products In Mexico:
The following are the popular nutraceutical products in Mexico:
- Omega-3 supplements.
- Weight gain supplements.
- Vit-C and Multivitamins.
- Calcium and Iron supplements.
- Herbal teas and powders.
Key Differences Of Nutraceutical Regulations In Mexico In Comparison To Other Countries:
- COFEPRIS demands pre-approval notification which is not mandatory in other countries.
- Labeling must be compulsorily in Spanish, whereas in other countries English is preferred.
- CBD or Cannabidiol is not allowed in nutraceuticals in Mexico.
Future Scope Of Nutraceuticals:
Mexican market holds a great scope for nutraceuticals as the people in Mexico are shifting towards a health-conscious approach and hence the demand for nutraceuticals is on a high rise. This serves as a great opportunity for local as well as foreign manufacturers to launch their products in Mexico.
We at Artixio, aim to help the manufacturers launch their nutraceutical products in the Mexican market successfully, helping you navigate all the regulatory compliance with our well-experienced local team with their regulatory intelligence-driven solutions. We promise to support you with your nutraceutical, right from the pre-registration compliance to the post-marketing compliance.
For more information or regulatory assistance, write to us at info@artixio.com or fill out our Contact Us form.
FAQs:
Are the nutraceuticals required to be registered before their sale in Mexico?
Yes, all nutraceuticals are required to register their product with the regulatory authority of Mexico that is COFEPRIS before its sale in Mexico.
Can foreign companies sell their nutraceuticals in Mexico?
Yes, they can sell their nutraceutical in Mexico, but they should comply with the Mexican regulations for nutraceuticals and should register their product with the COFEPRIS before its sale.
How can the manufacturer ensure its nutraceutical compliance in Mexico?
The manufacturer can ensure its nutraceutical compliance by appointing a Mexican local representative to oversee the regulatory process and find any regulatory step that is required to be fulfilled.
Is there any fee associated with the nutraceutical registration in Mexico?
Yes, the fee associated with the registration process of nutraceutical is around 215 USD.




