Nutraceutical products are seeing steady demand, and many food manufacturers are now looking to enter this segment. At the same time, FSSAI nutraceutical regulations in India need to be understood before moving ahead, since these products are not treated the same as regular food.
In the following sections, we’ll look at how nutraceuticals are regulated in India and what the FSSAI registration process involves.
Nutraceutical Regulatory Authorities In India
In India, nutraceuticals fall under FSSAI. The same authority that handles food safety also covers these products. So when you apply for registration, most of the rules you’ll be dealing with are from FSSAI itself.
If this is something you’re working on, we also take up nutraceutical product regulatory services in India.
Health supplement Product classification India
- Dietary Supplements: Products to enhance the diet with extra nutrients.
- Functional Foods: Offer health benefits beyond basic nutrition.
- Nutritionally Fortified Foods: Enriched with additional nutrients.
- Herbal & Ayurvedic Nutraceuticals: Derived from traditional ingredients, following Ayurvedic principles.
- Probiotics & Prebiotics: Live microorganisms for gut health.
- Sports Nutrition: Supports athletes with supplements and energy products.
- Medical Foods: Formulated for specific medical dietary management.
Nutraceutical product Registration process in India
- Application Submission: Submit an online application through the FSSAI portal, by following the steps given below:
1. The applicant should visit the official website FoSCoS and then click on the apply for new license/registration as shown in the screenshot below:

2. The applicant should select the premise of operation such as general, railway station, airport/seaport or tatkal registrations as shown below in the screenshot.

a) Then the applicant is asked to select the name of the state where the premises is located, he should select the appropriate state of his premise.

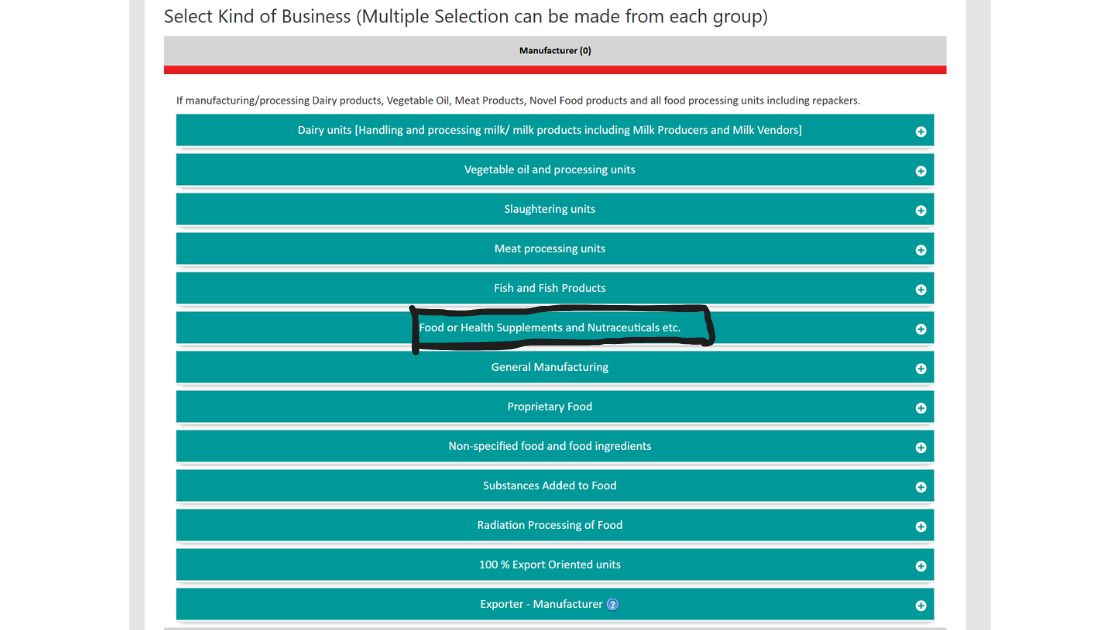
3. After selecting the state, as shown in the screenshot below select the kind of business.

4. Select the manufacturer option and then in select the Food or Health Supplements and select the type of license such as central as shown in the screenshot below.
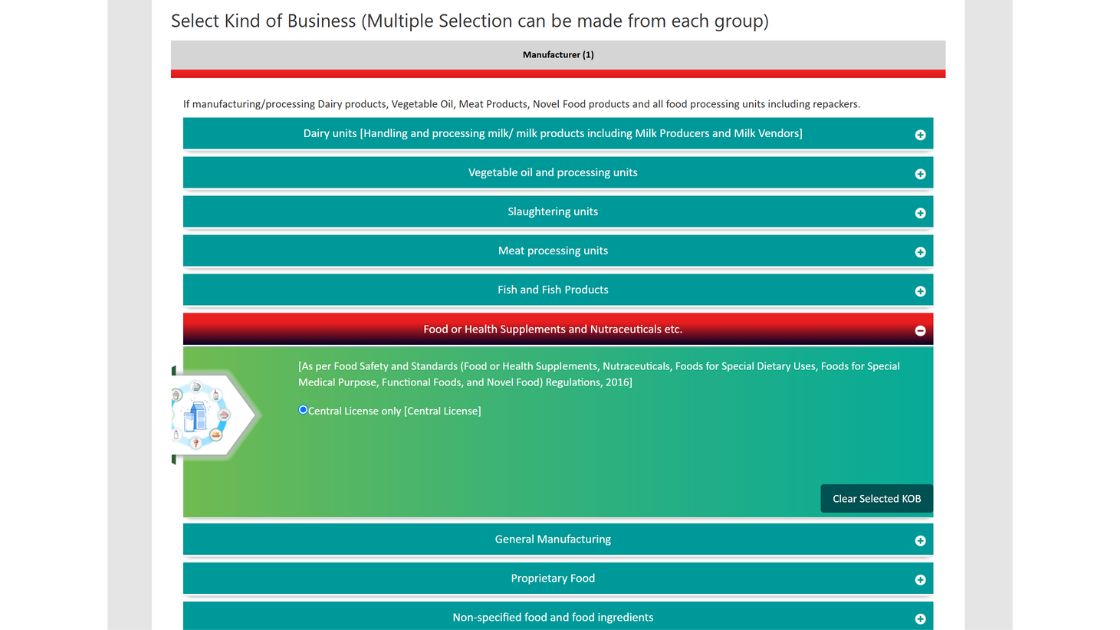
5. After proceeding, the next page will show for your eligibility for license, check it and click on the proceed button.
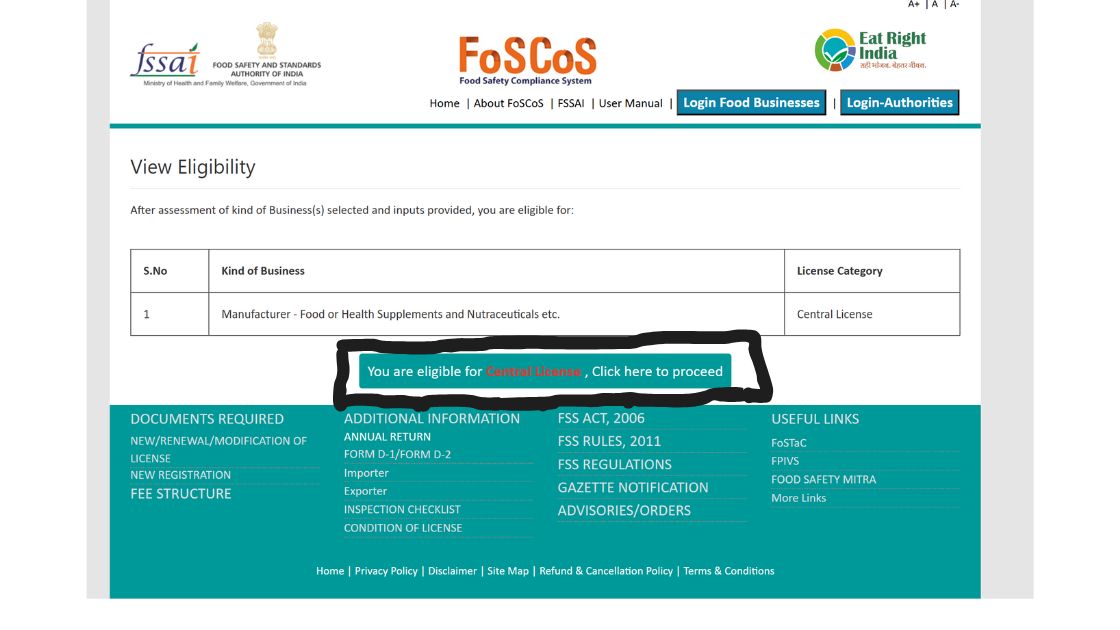
6. Then fill in all the Form B as per the details required, in the parts of the form such as premises, product selection, communication details, required documents and payment. Then finally after saving all the information submit your application.

- Product Details: Provide comprehensive details about the nutraceutical product, including its ingredients, formulation, intended use, and safety data.
- Safety Assessment: Furnish scientific evidence demonstrating the safety and efficacy of the product, especially for novel ingredients.
- Label Approval: Ensure that the product label adheres to FSSAI’s Nutraceutical labelling regulations, including accurate nutritional information and claims.
- Manufacturing Facility Details: Provide information about the manufacturing facility, equipment, and quality control measures.
- Ingredients Source: Document the source and quality of ingredients used in the product.
- Packaging Information: Submit packaging details that comply with FSSAI standards.
- Review and Approval: FSSAI reviews the application, assesses the product’s safety and efficacy, and grants registration if requirements are met.
- Compliance and Surveillance: After approval, maintain compliance with ongoing surveillance and audits by FSSAI to ensure product quality and safety.
Document Required for Nutraceutical Registration by FSSAI
- Product formulation and manufacturing process details.
- Proof of safety and efficacy.
- Lab analysis reports of the product.
- Details of the manufacturing facility and equipment.
- Proof of source and quality of ingredients.
- Packaging and labeling information.
- Declaration of conformity to FSSAI regulations.
Health supplement Labelling requirement
The label of a nutraceutical product should comply with FSSAI’s labeling regulations, which include information about.
- Product name.
- List of ingredients and allergens.
- Nutritional information.
- Manufacturing and expiry dates.
- Usage instructions.
- Country of origin.
- FSSAI license/registration number.
Nutraceutical Quality Control: FSSAI enforces quality control measures to ensure that nutraceutical products meet the specified standards and are safe for consumption.
Monitoring and Surveillance: FSSAI monitors the nutraceutical market to identify potential safety issues, misleading claims, and non-compliance with regulations. It takes corrective actions when necessary to protect public health.
Health supplements Import and Export
Imported nutraceutical products must also comply with FSSAI regulations. The importer needs to have a valid Importer Exporter Code (IEC) and obtain an FSSAI import license.
Fee structure and timeline associated with nutraceutical registration in India
Applicants should furnish a dossier with a complete application form and government fees of Rs. 50,000. The Food Authority will convene an expert committee for preliminary scrutiny and claim approval.
The timeline for obtaining the FSSAI Advertisement & Claims Approval for Food Supplements & Nutraceuticals is approximately one to eight days.
Conclusion
If there’s one thing the last few years have shown, it’s that people in India are more open to supplements than ever before. With that demand comes a responsibility to follow the rules that protect consumers. FSSAI’s framework may feel layered, but it’s there for a reason. Companies that spend a little extra time making sure their formulations, claims, and paperwork line up with the regulations generally move forward without major roadblocks. In a market that’s becoming crowded, doing things the right way has become a competitive advantage by itself.
For brands looking for end-to-end support with nutrition and supplement compliance in India, contact us at info@artixio.com
FAQs:
Can the nutraceuticals be sold through online mode in India?
Yes, a nutraceutical that complies with all the standards set in by FSSAI can be sold by online mode in India.
Does FSSAI non-compliance causes any penalties?
Yes, FSSAI non-compliance leads to penalties such as license cancellation, fines, recall, imprisonment of the products, etc.
What are the licenses required for nutraceutical registration in India?
The licenses such as FSSAI license, central or state license for nutraceutical registrations are required in India.
Can nutraceuticals in India make health claims?
Yes, a nutraceutical can make health claims if it has scientific data to prove it.
Do nutraceuticals require clinical trials conduction?
No, it is not mandatory for the nutraceuticals to carry clinical trials. However, if the nutraceutical makes any health claims the FSSAI requires clinical data to prove it.




