India’s cosmetic market is bustling with opportunities, attracting both domestic and international players. However, entering this lucrative market requires meticulous navigation through regulatory frameworks overseen by the Central Drugs Standards Control Organization (CDSCO).
CDSCO Cosmetics Registration in India for Importation
The importation of cosmetics from overseas into India falls under the jurisdiction of the Central Licensing Authority (CLA), the primary regulatory body responsible for enforcing the Cosmetic Rules of 2020. The Overseas Cosmetics shall have an active and valid Registration Certificate in place to be imported into India. For importation, CDSCO registration of cosmetics in India as well as their manufacturing sites is a mandatory pre-requisite.
Who Can apply for CDSCO Registration Certificate for import of Cosmetic Products?
Entities eligible to apply for CDSCO registration of cosmetics in India include importers, Indian authorized agents, foreign enterprises with Indian subsidiaries, and corporations. Authorization from the foreign manufacturer, duly authenticated, is a prerequisite for submission. The manufacturer’s authorization to their agent in India must be properly authenticated, either before a first-class Magistrate in India or by the competent authority in the country of origin as per its laws, or by an authority specified in the First Schedule.
Also Read: CDSCO Registration Process for New Cosmetics
How can one apply for CDSCO Registration Certificate for import of Cosmetic Products?
The CDSCO registration of cosmetics in India begins with submitting Form COS-1 through the CDSCO online portal. Alongside the application, a set of documents, including ingredient lists, labels, manufacturing licenses, and non-animal testing declarations, must be furnished. The documents that must be submitted for Registration Certificate for import of cosmetic products include –
- Covering Letter mentioning the purpose of application
- Authorisation from the manufacturer (duly authenticated)
- List of Ingredients along with its percentage contents Labels of proposed products
- Specifications and testing methods Pack insert
- Manufacturing Licenses/undertaking for no provision of manufacturing license in the country.
- Free Sale Certificate (duly authenticated)
- Non-Animal Testing Declaration
- Declaration for Heavy Metal and Hexachlorophene content.
- Other documents (If any).
- Application in Form COS-1
- Fee: Original Bharatkosh e-receipt for fee paid.
Also Read: CDSCO Labeling Requirements for Cosmetics in India
Importation of Ready to fill Cosmetic Bulk Finished Formulations into India
The importation of ready-to-fill bulk cosmetic finished formulations into India is sometimes preferred for cost-effectiveness and operational efficiency. In such instances, the application process includes providing details regarding the registered brand owner of the finished product in India along with a copy of a valid manufacturing license for the finished formulation. This license must be obtained from the State Licensing Authority and is necessary for the cosmetic to be filled in its finished form.
Exemptions for import of cosmetic products in to India without CDSCO Registration Certificate
- Certain categories of cosmetics are eligible for import into India without requiring a registration certificate issued by CDSCO, provided they are not intended for domestic sale. These exemptions include:
- Cosmetics in bulk intended for repackaging solely for 100% export to other countries. In such scenarios, the importer must secure requisite permission from CDSCO, HQ New Delhi, and provide a written undertaking affirming that these products will not be sold domestically.
- Cosmetics earmarked for research and development purposes, such as packaging trials, consumer studies, shelf-life studies, and transport studies. A No Objection Certificate must be acquired from the respective Port offices of CDSCO.
- Cosmetics designated for sale exclusively to overseas passengers at duty-free shops located within international airports.
- Cosmetics included in amenity kits intended for the exclusive use of international passengers on a complimentary basis, provided they are not intended for domestic sale.
- Cosmetics imported for use in hotels for their internal consumption, subject to providing a notarized undertaking confirming this purpose.
- Hair fixers, specifically mucilaginous preparations containing gums, intended for use by men for beard fixing.
Post-approval Changes in the CDSCO Registration Certificate for import of cosmetic products
Once the Central Licensing Authority (CLA) issues the Registration Certificate for import of cosmetics in to India under COS-2, applicants has the flexibility to implement any necessary changes. In case the However, the nature of these changes determines the course of action and it is crucial to understand when a fresh registration is required versus when mere notification suffices.
Any alteration in the constitution of the registration holder or overseas manufacturer necessitates a fresh registration within 180 days of the change. It’s noteworthy that the existing registration certificate remains valid until the CLA either issues a new certificate or rejects the application for amendment. If an applicant has submitted an application for a Registration Certificate but it has not yet been issued, and there is a change in the constitution of either the Manufacturer or Indian Agent, or the address of the manufacturer, the applicant shall submit fresh application. However, such application need not be accompanied by any fees.
Conversely, changes such as modifications in product labeling, composition, or testing specifications do not require fresh registration; instead, they warrant notification to the Central Licensing Authority within 15 days, accompanied by an undertaking of compliance with Bureau of Indian Standards (BIS) standards.
Similarly, alterations in the name or address of the registration certificate holder or overseas manufacturer require an application for amendment within 60 days from the date of such changes. Additionally, any changes in the name and/or address of the Indian agent/Importer/legal or actual manufacturer, or a change in constitution following the issuance of the registration certificate, must be promptly communicated to the Licensing Authority. It’s vital for industry stakeholders to grasp these intricacies to ensure regulatory compliance and smooth operations.
A corporate acquisition or merger involving one company absorbing another is regarded as a modification in the company’s structure. The Indian agent or importer is required to promptly notify the licensing authority in writing and submit a new application.
CDSCO timelines for issuing Registration Certificate for import of Cosmetic Products
The CDSCO review of applications made under COS-1 for Registration Certificate for cosmetic products may take a maximum of 180 days from date of submission. The CDSCO usually aims for a 90-day processing timeline for applications.
CDSCO Fees for Processing COS-1 Applications for import of Cosmetic Products
The fee structure applies to each cosmetic category and manufacturing site, with additional fees for each specified category of cosmetic and variant.
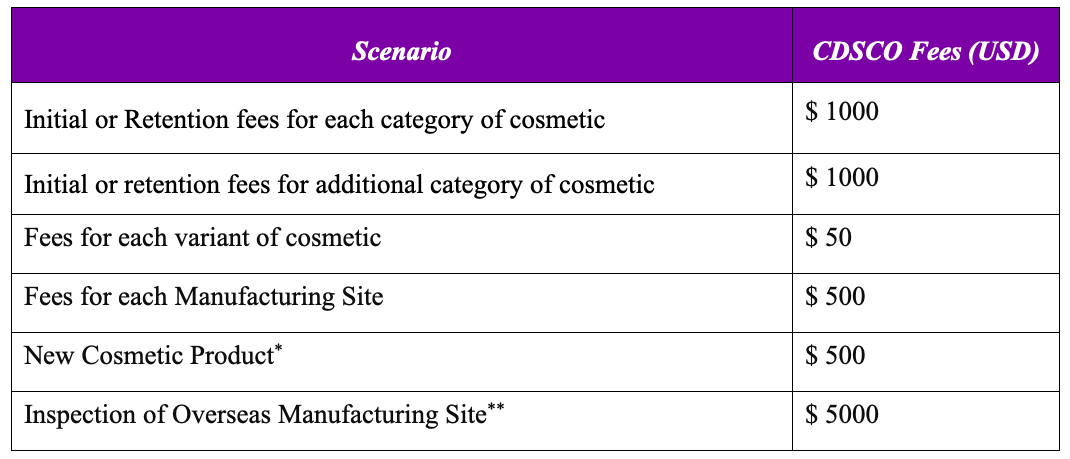
*New Cosmetic Product contains a novel ingredient which has not been used anywhere in the world or is not recognized for use in cosmetics in any National or International literature
** In case CDSCO identifies a need to inspect the overseas cosmetic manufacturing sites, the applicant shall pay additional fees for inspection upon communication from the Agency.
Validity of Registration certificate for import of cosmetic Products
The Registration Certificate remains valid indefinitely, provided the retention fee is paid every five (05) years. However, it may be subject to suspension or cancellation by the Licensing Authority. Failure to pay the retention fee within 180 days incurs a late fee of two percent per month, and if the fee remains unpaid during this period, the registration certificate will be considered canceled.
Understanding Regulatory Restrictions
Importing cosmetic products into India is governed by the Cosmetic Rules of 2020, enforced by the Central Licensing Authority (CLA). It’s crucial to note the restrictions imposed.
- Animal Testing: Cosmetics tested on animals are strictly prohibited for import.
- Shelf life of the imported cosmetics: Only products with a minimum expiry period of six (06) months from the date of import are permitted for importation into India.
- Ingredient Restrictions: Cosmetic Products containing certain ingredients, like hexachlorophene are prohibited from importing into India.
The process of importing cosmetic products into India is governed by regulatory measures enforced by the Central Drugs Standards Control Organization (CDSCO). From the initial application submission to post-approval compliance, meticulous attention to detail is essential. Understanding and adhering to these regulations are paramount for businesses seeking to tap into India’s lucrative cosmetic market.
Partner with Artixio for Expert Regulatory Support
Don’t let regulatory hurdles hinder your expansion into the vibrant Indian cosmetic market. If you’re planning to import a cosmetic product and the CDSCO steps feel a bit overwhelming, reach out anytime. We can walk you through the paperwork and help you get it right the first time. Contact Artixio today to unlock opportunities and propel your business forward.




