CDSCO Medical Device Regulatory Services in India
Artixio is a Leading Medical Device Regulatory Consultant in India, your Partner for End-to-End Regulatory affairs Services in India, From Strategy to Device Registration and Post Approval Maintenance.
India-Based Experts for Medical Device Registration
Medical Device Regulatory Consultants in India for IVD and Market Access: Artixio offers extensive regulatory affairs services to global manufacturers, facilitating smooth navigation of India’s CDSCO regulations.
Artixio helps companies handle product approvals for medical devices and IVDs in India. From CDSCO submissions to in-country representation, our team works directly with clients to meet Indian compliance needs..
- Expert Medical device registration services in India
- IVD registration and market access and documentation
- Guidance on medical device control division processes
- India-based medical equipment consulting
CDSCO coordination and classification support
The Central Drugs Standard Control Organization (CDSCO) regulates the registration of medical devices in India. The registration makes sure that the devices meet safety and performance standards before they can be marketed.
Our Services

Regulatory Intelligence & Strategy

MDR Gap Assessment

Medical Device Regulatory Affairs

Quality Consulting

Technical and Medical Writing

Clinical & Performance Evaluation

Medical Device Registration / Certification

Authorized Agent / Legal Representation

Post-Market Surveillance
Key Regulations for Medical Devices in India
Classification of Medical devices In India
Medical Devices is categorized into four risk categories: A, B, C, and D. The CDSCO processing fees for the application are determined by the device Class. The process takes 6 to 9 months regardless of the Class.
Artixio offers expert-led medical device regulatory services in India, covering documentation, classification, and CDSCO submission. Our local team helps ensure smooth product approvals and compliance with Indian standards.
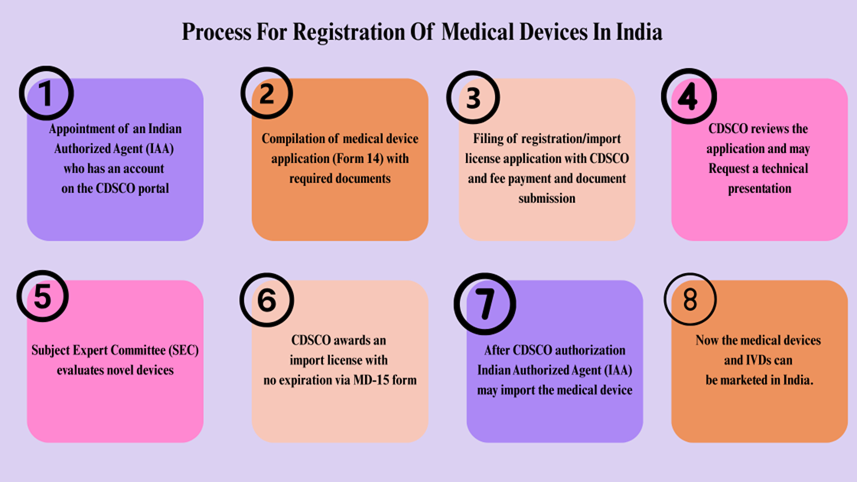
Process for Registration of Medical Devices in India:
Documents Required for Medical Device Registration In India
- Form 40
- TR6 Challan
- Power of Attorney
- Schedule D(I)
- ISO 13485 Certificate
- Quality Assurance Certificate
- CE Certificate
- Declaration of Conformity
- Free Sale Certificate
- Post Marketing Study Reports
- Plant Master File
- Device Master File
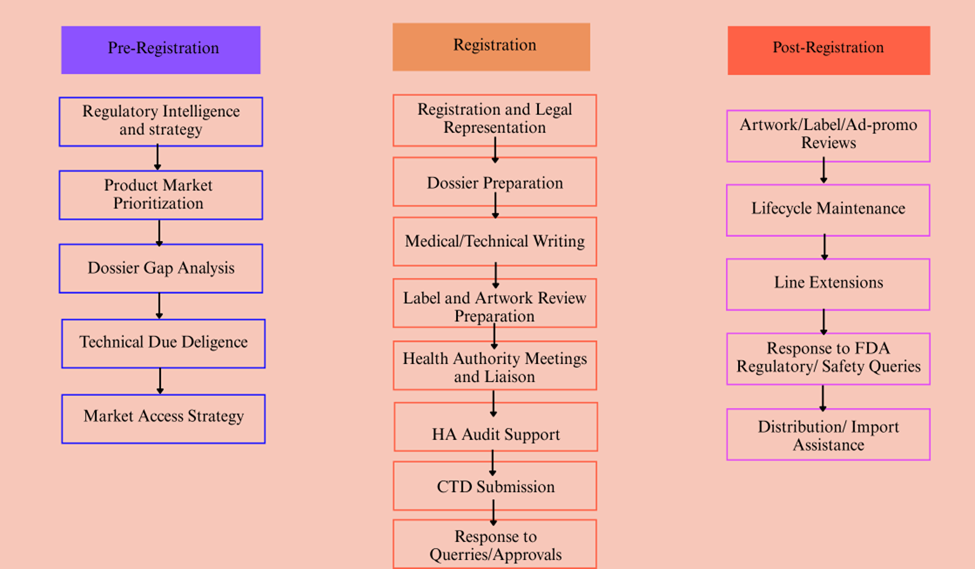
Artixio’s Medical Devices Regulatory Compliance Services in India
Our Regulatory and Market Access experts based in India, coupled with the Global Regulatory Projects team, have helped over a hundred pharmaceutical, medical devices, diagnostics, cosmetics, nutraceuticals, supplements, and veterinary products successfully register and maintain compliance in India. Combined with Artixio’s intelligence-based services platform, QuriousRI, innovators and manufacturers from around the world have benefited by seamlessly managing the regulatory strategy across the product development and commercialization value chain.
For Medical Devices, from strategic planning and registration to post-market compliance, Artixio provides high-quality medical device regulatory affairs services in India to support your business needs.
IVD Regulatory Affairs and Medical Equipment Consulting in India
In India, IVD products and medical equipment must meet CDSCO’s standards before reaching the market. Artixio helps you make sense of IVD regulatory requirements, prepares the needed documents, and provides on-ground support. With our experience in India’s IVD approvals and consulting for medical equipment, your path to compliance becomes simpler.
Full-Scope Regulatory Services for Market Entry in India
Why Choose Artixio for Medical Device Approval in India



















FAQS
1. Who act as the Central Licensing Authority for Medical Devices in India?
2. What is the mode of submission of various applications in MDR-2017 to the Licensing Authority?
3. If a device is complying with ISO/IEC standards, would it still need to follow BIS standards?
4. What are the Labeling requirements for Medical Devices in India?
5. Is a license is required for import of medical device for marketing in India?
6. Who is an “Authorized agent” for import of medical devices in the country?
7. What is the timeline for the grant of Import license in Form MD-15?
Related India Services
Regulatory Expertise Across
Multiple Countries












Insights from Artixio - Tips & Articles

CDSCO Simplifies Subsequent Importer
India’s regulatory authority, the Central Drugs Standard Control Organization (CDSCO), has published an important...

CDSCO Medical Device Approval
Central Drug Standard Control Organization (CDSCO) is the regulatory authority that regulates Medical Devices...

IVD Medical Devices Registration
IVDs in India are regulated as drugs under Section 3(b)(i) and 3(b)(iv) of the...






















