Many companies entering Mexico underestimate how detailed the pharmaceutical regulations in Mexico can be. What looks like a standard submission on paper often turns into multiple clarification rounds once it reaches Comisión Federal para la Protección contra Riesgos Sanitarios (COFEPRIS).
It’s important to understand the COFEPRIS registration process for pharma products in a practical way, not just from a regulatory checklist perspective. . In this guide, we’ll walk through how the process works, what needs to be prepared, and where most applications tend to slow down.
COFEPRIS Role in Pharmaceutical Regulations in Mexico
COFEPRIS is involved at almost every stage of Pharmaceutical products registration in Mexico. COFEPRIS reviews applications and decides whether a product can be approved in Mexico. It also looks over the post marketing surveillance and GMP compliance of the pharmaceutical product after its registration. Because of this, working within pharmaceutical regulations in Mexico is not just about submission—it’s about staying compliant even after approval.
Classification Of Drugs According To COFEPRIS:
COFEPRIS classifies drugs on the basis of preparation mode which categorizes drugs as:

- Magisterial Drugs: These are the ones which are prepared as per doctor’s prescription
- Officinal Drugs: These are as per the Mexican Pharmacopoeia
- Pharmaceutical Specialties: These are the ones prepared with formulas authorized by the Ministry of Health, in collaboration with the pharmaceutical industry
Further it is classified on based of origin:
- Allopathic: Any substance or mixture of substances of natural or synthetic origin that has a therapeutic, preventive or rehabilitative effect, that is presented in a pharmaceutical form ‘and is identified as such by its pharmacological activity, physical, chemical and biological characteristics, and is registered in the Pharmacopoeia of the United Mexican States for allopathic medicines
- Herbal: Products made from plant material or a derivative thereof, whose main ingredient is the aerial or underground part of a plant or extracts and tinctures, as well as juices, resins, fatty and essential oils, presented in pharmaceutical form, whose therapeutic efficacy and security has been scientifically confirmed in national or international literature.
- Homeopathic: Any substance or mixture of substances of natural or synthetic origin that has a therapeutic, preventive or rehabilitative effect and that is prepared in accordance with the manufacturing procedures described in the Homeopathic Pharmacopoeia of the United Mexican States, in those of other countries or other national and international sources of scientific information.
The third classification is based upon the mode of delivery for products; it is further divided into six categories:
- Medicine that needs prescription or special permission issued by MoH
- Medicines which need prescription, and every sale should be recorded and can be filled up to two times
- Medicines which can be obtained only with prescriptions and can be filled up to three times
- Medicines that require prescriptions but can be refilled multiple times
- Non-Prescription drugs for sale in pharmacies
- Medicines that don’t require prescriptions and cane be sold anywhere other than pharmacies.
Pharmaceutical companies planning market entry in Mexico may benefit from experienced regulatory support. Discover how Artixio assists with COFEPRIS pharmaceutical compliance, registrations and ongoing regulatory requirements.
Submission Dossier Preparation:
The layout of the submission dossier depends on the type of product intended to be registered, such as a new molecule, generic, orphan drugs, etc. The format for various product categories is given below:
1. New Molecules:
Module I. Legal/Administrative information
Module II. Quality information
Module III. Preclinical studies
Module IV. Clinical studies
2.Generic drugs:
Module I. Legal/Administrative information
Module II. Quality information
Module III. Bioavailability and/or bioequivalence
3.Vaccines:
Module I. Legal/Administrative information
Module II. Quality information
Module III. Preclinical studies
Module IV. Clinical studies
4.Orphan drugs:
Module I. Legal/Administrative information
Module II. Quality information
Module III. Justification of ‘orphan drug’ status
Module IV. Preclinical studies
Module V. Clinical studies
Pharma Products Registration Process with COFEPRIS
The process of registration of COFEPRIS begins with the consultation meeting with COFEPRIS followed by the GMP inspection held by COFEPRIS. Both COFEPRIS registration steps are explained in detailed below:
COFEPRIS Registration Process (Step-by-Step)
Pre-submission meeting with NMC
- Usually takes around 1–2 months to schedule and complete.
- Helps clarify what COFEPRIS expects before you prepare the full dossier.
- Not always mandatory, but useful for new molecules or complex products.
Dossier preparation
- Usually takes a few months, depending on how ready the data is.
- Everything needs to be arranged as per COFEPRIS format, including Spanish documents.
- If something is missed here, it usually shows up later during review.
GMP inspection request
- Timelines vary, but planning for a few months is safer.
- COFEPRIS may still ask for inspection details even if approvals exist in other countries.
- Site information should match what’s submitted in the dossier.
Submission (third party or direct route)
- Submission itself is quick, but acceptance can take a few weeks.
- Many companies use an authorized third party to speed up the initial screening.
- Incomplete submissions may not move forward to review.
Technical review & queries
- This is the longest stage — often around 8 to 12 months.
- Queries are common, even for well-prepared dossiers.
- Delays usually happen here, especially if responses take time or are unclear.
Approval & market authorization
- Timelines vary, but approval doesn’t always come immediately after review.
- Once approved, the product can be sold in Mexico.
- Compliance doesn’t stop here, some requirements continue even after approval.
Request GMP inspection from COFEPRIS
-
GMP inspection must be requested to COFEPRIS as Mexico abides to their own CMP practices and standards
-
GMP certificates for other country of origin is exempted
-
GMP certificate is must for all the manufacturing sites
Checklist documents for GMP Inspection
1. Name and general information of the establishment
2. Name of the drug or medicine for which you are requesting verification of GMP
3. Name and full address of the establishment(s) involved in each stage of manufacturing
4. Description of the process that is carried out in each of the establishments involved
5. The manufacturing process for which verification of GMP is requested
6. A list and description of products that are made
7. Name of the legal representative, health officer or person designated by the establishment to attend the diligence
8. Organization charts (general, of the production and quality departments, indicating the reporting lines)
9. Plans of the establishment and production areas
10. Block diagram of the manufacturing process
11. General summary of the quality system including validation and qualification
12. Information from the last two annual review reports, specifically indicating: manufactured lots, rejected lots (indicating reasons), released lots that were subject to investigation, conclusion and actions carried out, number of reprocessed batches, complaints, returns and withdrawal of products from the market, as well as conclusions of the report.
Prepare and Submit registration dossier to third party or directly to COFEPRIS for review and approval.
COFEPRIS Modules For Pharmaceutical Registration In Mexico
The COFEPRIS consists of four modules whose structure is shown below:
Module I: Legal/Administrative Information:
-
Application Form
-
Proof of Fee Payment of fee
-
Sanitary authorization (This is applicable for site located outside Mexico, one has to provide license, certificate or other document authorizing the site to the activities related to manufacturing of pharmaceutical products of interest, issued by the competent authority of the country of origin, legalized or apostilled, translated to Spanish by an official translator).
-
Responsible Sanitario notice
Labeling Information Required as per COFEPRIS
-
GMP certificate API manufacturing sites
-
GMP Certificate for Finished product manufacturing sites
-
Certificate of Pharmaceutical Product (This is applicable for drug manufactured outside Mexico)
-
Commercial name of products
-
Intellectual property/Patent Information
-
Information on the waiver for the local manufacturing facility requirement.
Module II: Quality Information
-
API: Manufacturing information, General Information, Quality Control (Literature/Pharmacopeial references. Specifications. Analytical methods. Validation reports. Certificates of analysis (CoAs).
-
Excipients and additives: For new additives, usage safety information, Quality control ((Literature/Pharmacopeial references. Specifications. Analytical methods. Validation reports. Certificates of analysis (CoAs).
-
Finished product: Pharmaceutical development, Formulation/Manufacturing information, in process controls, Quality control, Monographs specifications, Analytical methods certificates of analysis, Stability studies and stability data, Packaging materials (description and capacity)
Module III: Preclinical Studies (as applicable for new molecules, Vaccines and Orphan Drugs)
-
Preclinical studies: Pharmacodynamic studies, Pharmacokinetic studies, Toxicology studies
Module IV: Clinical Studies- Phase I, Phase II, Phase III and Phase IV studies
Registration Process for Foreign Manufacturer:
The process would be different for organizations with prior registration in reference countries or without registration in reference countries
Prior registration in reference countries (USFDA, Health Canada, Swissmedic, EMA, TGA)
- Request meeting with COFEPRIS New Molecule Committee (NMC)
-
Using 3rd party for evaluation of technical files
-
3rd Party issues evaluated report to Manufacturer
-
Submit registration request to COFEPRIS
Common Challenges Encountered In COFEPRIS Registration
Some of the common challenges faced by the applicants who wish to register their products with COFEPRIS is as follows:
Complex Regulatory Requirements:
The COFEPRIS has a detailed complex regulatory requirement which might sometimes become difficult to follow. This can be overcome by being updated with the latest new updates and hiring a local experienced representative in Mexico.
Language Barrier:
As the complete regulatory procedure in Mexico is in Spanish language it may create a barrier for the foreign people who wish to register their products in Mexico. This problem can be overcome by correctly translating all the requirements.
COFEPRIS Timelines:The review time for COFEPRIS registration might be long due to many registrations handled by COFEPRIS simultaneously. This can be overcome by ensure a correct and authentic registration application to avoid additional non-compliance correction timelines.
High Costs of the Registration Fees:
The complete process of pharmaceutical product registration with COFEPRIS can be very costly for the applicant to afford sometimes, hence the applicant should plan out the budget first and then only register with COFEPRIS.
COFEPRIS Registration Fees:
The COFEPRIS registration fees depends on the type of product, its complexity and the according to the risk associated with it. However, the COFEPRIS registration fees are high therefore one should plan its budget before registering with COFEPRIS and stay updated with the latest changes in the fee structure.
The table below explains the approximate registration fee for various products:
| PRODUCT TYPE | REGISTRATION FEES |
| Generic Drugs | MXN 82,012 approx. |
| New Molecule | MXN 146,642 approx. |
| Class I Medical Devices | MXN 13,000 approx. |
| Class II Medical Devices | MXN 20,000 approx. |
| Class III Medical Devices | MXN 25,000 approx. |
Timelines by COFEPRIS For Pharma Product Approvals
The response time for New Molecules and Generics is anywhere around 180 days whereas requesting a meeting with COFEPRIS New Molecule committee takes around 60 days and another 20-40 days for receiving New Molecule committee conclusions after meeting.
At Artixio, we have a team of experts who understand the Mexican market and comes with decades of experience working with the authorities in Mexico. Our team has helped numerous pharmaceutical companies complying to COFEPRIS’s regulatory needs. Our experts have expertise in providing end to end solution for Mexican registration requirements:
-
Preparation and submission of dossier, complying to necessary guidelines to ensure a smooth process of submission for pharmaceutical products
-
We have a team of consultants who have experiences handling GMP audits for manufacturing sites updating system as per GMP requirements
-
Artixio team can partner as your authorized legal representative for Mexico helping with health authority (COFEPRIS) communication on a regular basis
-
We have our partners who can also assist you with local clinical and non-Clinical testing of your products at ISO compliant laboratories in Mexico
Recent updates in COFEPRIS pharma registration guidelines
COFEPRIS Changes to the Drug Registration Process in Mexico (2023 update)
There are some updates came in around 2023 for the COFEPRIS Drug registration process. The overall system stayed the same, but a few steps in how submissions move forward were changed.
- One difference is the alignment with ICH. If you’ve worked on US or EU filings, some parts will feel familiar now, though Mexico still has its own expectations in terms of documentation.
- There’s also more room for discussion before submission. Earlier, most of the clarity came only after filing, but now there’s a bit more flexibility to engage beforehand and avoid obvious gaps.
- Internally, the review flow seems more streamlined compared to before. Instead of too many back-and-forth loops, it’s a bit more contained, although timelines still depend a lot on how clean the dossier is.
- Typical timelines have come down in some cases, but it’s not something you can rely on unless the submission is solid. Incomplete or inconsistent data still leads to delays.
- On the documentation side, nothing very unusual was introduced. You still need the core set — application details, clinical data, manufacturing information, and labeling. That part hasn’t really changed much.
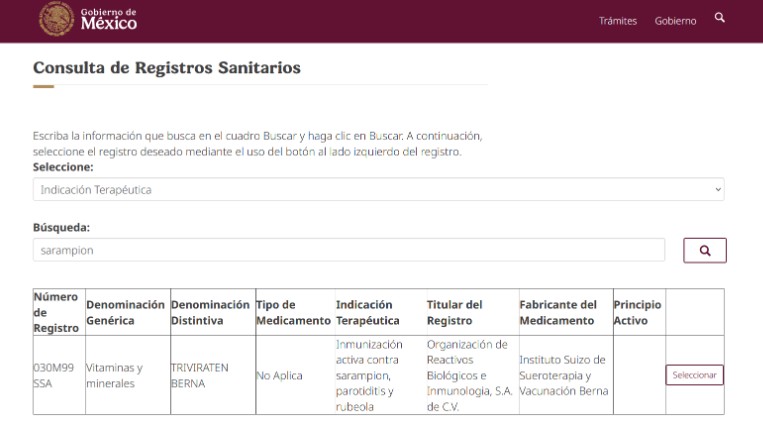
January 2026 Update: Public Access to Pharmaceutical Premarket Authorizations
It is possible to review current premarket authorizations for medication here:
The site can be searched by:
- Premarket authorization
- Generic name
- Distinctive name
- Type of medication
- Therapeutic use
- Name of premarket authorization holder
- Name of medication manufacturer
- Active ingredient
I chose for the example “therapeutic use” and added “Measles”, and got what follows:
If “Seleccionar” is chosen on the site from this example, then the full information is displayed.
Conclusion
The COFEPRIS registration process for pharma products can take time, especially if the submission isn’t fully aligned with pharmaceutical regulations in Mexico. Most of the problems don’t show up early, they come during the review stage.
If you need support with Mexico registrations, the Artixio provides Pharmaceutical Product regulatory affairs in Mexico and our team works closely on submissions, compliance, and ongoing coordination with COFEPRIS. Connect with us at info@artixio.com
FAQs:
Why is COFEPRIS important for pharmaceutical registration in Mexico?
COFEPRIS is important as it is the regulatory authority for Pharma products in Mexico. You need COFEPRIS approval to sell a pharma product in Mexico. Without it, the product can’t enter the market at all.
Can a pharmaceutical product be registered in Mexico without COFEPRIS registration?
No. It has to go through COFEPRIS first. Otherwise, it won’t be allowed in the market.
How can the COFEPRIS registration process be tracked?
The COFEPRIS registration process can be tracked through its online portal.
Can a pharmaceutical product be advertised before registration approval from COEFPRIS?
No, a pharmaceutical product cannot be advertised before receiving a registration approval from COFEPRIS and doing so may cause legal consequences.