- Home
- >
- Quatar
- >
- Pharma
- >
- Regulatory Affairs
Pharmaceutical Regulatory Affairs in Qatar
Artixio covers all, from the early stage regulatory strategy to product registration globally, efficient regulatory operations and then post marketing maintenance in Qatar.

Services We Offer
Regulatory Affairs Consulting for Pharmaceuticals in Qatar
Artixio has established collaborations with 50+ global pharmaceutical firms, providing successful market entries and regulatory approvals for a wide range of products. We have a team of experts with more than 30 years of experience with respect to Qatar’s regulatory framework and has delivered high impact solutions to multiple therapeutic areas and innovations.
We provide support from early stage development via product registration to post approval support. W provide integrated and comprehensive regulatory solutions using regulatory intelligence. Our services include strategic consulting, regulatory writing, dossier preparation, electronic publishing, labelling and artwork management, promotional material review, and regulatory information management.
Pharmaceutical Regulatory Authorities in Qatar
| Regulatory Body | Role |
|---|---|
| Ministry of Public Health (MOPH) | Monitors all regulations related to health, including pharmaceutical products. |
| Qatar Drug Control Department (DCD) | Responsible for drug registration, distribution, and licensing of pharmaceutical products. |
| Qatar Standards Organization (QSO) | Ensures compliance with quality standards for imported pharmaceutical products. |
Pharmaceutical Regulations in Qatar (MoPH)
The central regulatory body of Qatar is Qatar Ministry of Public Health (MoPH) which regulates the process of drug registration and approval. It ensures that all the products meet the standards for efficacy, safety and quality.
Major Regulatory Requirements
The applicant must fulfil these major requirements for submitting a drug product application-
- Valid and legal Good Manufacturing Practice (GMP) certificate from the origin country’s health authority.
- Valid and legal manufacturing license (ML) from the origin country’s health authority.
- A relationship letter between the MAH and manufacturer
- The product list which are manufactured by the manufacturer listing their trade and generic names and pharmacological groups.
- Site master file copy
- If available, then certificate of registration of the manufacturing sit in gulf countries.
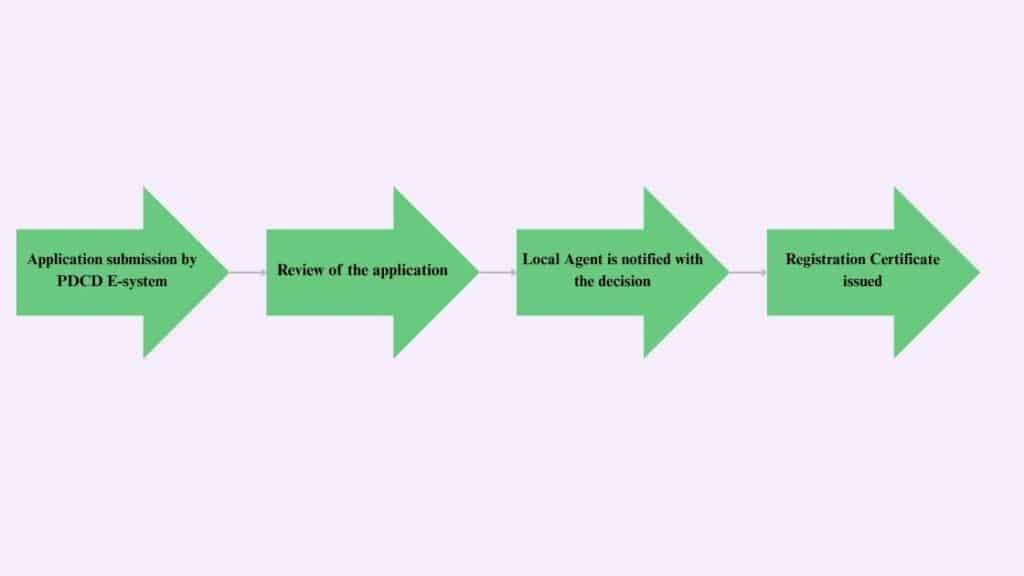
Regulatory Process Overview
The applicant must fulfil these major requirements for submitting a drug product application-
The application is submitted through online portal PDCD E-system.
The application is then assessed and for any further requirements the local agent is informed.
The file is then presented to the registration committee and the decision is notified to the local agent.
If the decision is positive, then the registration certificate is generated online on the official website.
Pharma Regulatory Affairs Services in Qatar
Artixio offers a range of regulatory affairs solutions in Qatar to assist pharmaceutical companies with product registration:
- Medical & Technical Writing
- Due Diligence and Gap Analysis
- Dossier Preparation and Compilation
- Dossier Publishing & Submission
- Product Registration
- Marketing Authorization Holder (MAH / LR)
- Investigational New Drug (IND) / Clinical Trial Application (CTA)
- New Drug Application (NDA) / Marketing Authorization Application (MAA)
- Post-Approval Lifecycle Maintenance, Artwork & Label Review
- Advertising & Promotional Material Review
- Distribution and Import Assistance
Pharmaceutical Regulatory Process (MoPH) in Qatar
Marketing Authorization Holder Responsibilities
- Ensuring safety, efficacy and quality of the product throughout its lifecycle.
- Preparation of the dossier in eCTD format.
- If any product variation is there they have to inform the PDCD.
Local Agent Role
- They have to handle the communications from MoPH.
- The person or company must be a Qatari registered organization meeting all the requirements of MoPH.
- Responsible for the registration of MAH and the manufacturing site before the submission of the application.
Submission Process
- All the applications have to be submitted through PDCD system.
New Product Registration
Online Application Filling
- The applicant should be complete and submitted through the PDCD E-system.
- Firstly, the application has to go through the preliminary review and then for file submission.
Online Application Filling
- The applicant has to submit all the required documents. The authority will check that all the requirements are met and proceed for the review.
Review Phase
- The application will go through the assessment. If any query arrives it will be notified to the applicant through the local agent.
- If the PDCD requirements are not met within 4 months, the application will be rejected automatically.
Application Acceptance
- After the validation, assessment and if all the MoPH requirements are met then the file will be submitted to committee and they will give the decision.
Labelling Requirements
- The compliance should be there with Qatari labelling and packaging requirements.
- All the information regarding the drug’s packaging, dosage, warnings and expiry date.
Why Choose Artixio for Pharmaceutical Regulatory Affairs in Qatar






FAQs
What is the timeline for the registration certificate?
The registration certificate is valid only for 5 years. The renewal request must be submitted before 6 months from the date of expiry.
Is there a fast track review for GCC-centrally registered products?
PDCD IN Qatar provides fast track review for the new products already registered in GCC. This process generally eliminates the full evaluation of the application but some requirements should be met.
What are the major requirements for fast tracking the GCC-centrally registered products?
- A valid GCC registration certificate must be there.
- Any variations after the registration during initial period must be submitted.
Do I need local representation for pharmaceutical registration in Qatar?
Still Have Questions ?
Specialized Regulatory Affairs
Services Across Multiple Industries
Expert Regulatory Services To Streamline Compliance

Regulatory Intelligence & Strategy

Medical & Technical Writing

Publishing & Submission






Regulatory Affairs Across Multiple Countries

India

Singapore

Mexico

Brazil

Vietnam

Malaysia

Argentina

Colombia

Taiwan

China

China

Thailand

Indonesia

Philippines

USA

Japan

Qatar

South Korea
Insights from Artixio - Tips & Articles

Cell and Gene Therapy
Cell and Gene Therapy Products are modern treatments to treat diseases. Treatment of these...

Cosmetics Regulations & Registration
Germany is one of the biggest cosmetics markets in Europe, but entering the market...

Why Medical Device Submissions
Introducing a new medical device to the international market requires that you not only...

