- Home
- >
- Singapore
- >
- Biologics
Biologics Product Registration and Regulatory Services in Singapore
A trusted End-to-End Service Provider for Biologics in Singapore. We support biologics product registration in Singapore, helping you meet HSA’s regulatory standards with clear documentation, timely submissions, and expert guidance at every step.
Regulatory Support for Biologics Product Approval in Singapore
Artixio provides complete end-to-end support for registration of a biological product with Health Sciences Authority (HSA), a regulatory authority of Singapore. Our consultants in Singapore are highly experienced in product commercialization and lifecycle management. Our regulatory strategy helps clients to maximize commercial potential by reducing the time to market entry.
Biologics Product Regulatory Solutions in Singapore
Applicants can submit Therapeutic Product applications through the following two types
- New Drug Application (NDA-1, NDA-2, NDA-3)
- Generic Drug Application (GDA-1, GDA-2)
Biologics Product Registration and Regulatory Services in Singapore
Registration of a Therapeutic product in Singapore takes place through following steps
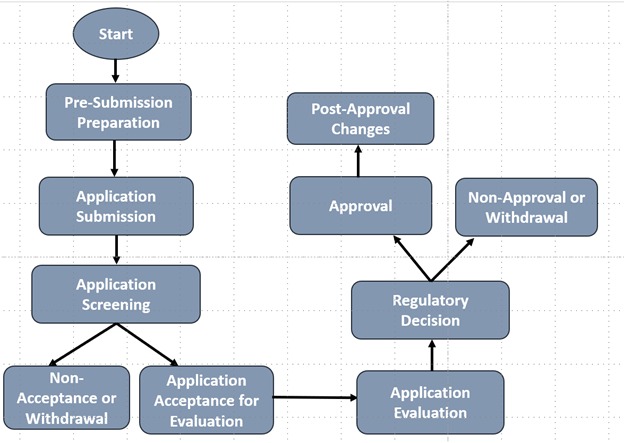
The application dossier must be organized in either ICH-CTD or ASEAN-CTD format, structured as follows
| Documents | Location in ICH CTD | Location in ASEAN CTD |
|---|---|---|
| Administrative documents | Module 1 | Part I |
| Common Technical Document Overview and Summaries |
Module 2 | Incorporated in Parts II, III and IV |
| Quality Documents | Module 3 | Part II |
| Non-Clinical Documents | Module 4 | Part III |
| Clinical Documents | Module 5 | Part IV |
Artixio Provides the Following Services for Biologic Products in Singapore
- End-to-End services to commercialize the biological products in singapore
- Provide the effective Product Strategy
- Pre-submission preparation
- Dossier preparation and submission
- Providing response to Health Authority’s Queries
- Effective market access strategy
Why Choose Artixio for Biologics Product Regulatory Solutions in Singapore?






FAQs
How is the therapeutic product evaluated in Singapore?
- Full Evaluation (For new product that has not been approved by any drug regulatory authority, applicable to NDA)
- Abridged Evaluation (For new or generic product approved by at least one drug regulatory authority, applicable to NDA & GDA)
- Verification (For new or generic product approved by the reference drug regulatory agencies, applicable to NDA & GDA)
- Verification CECA (For generic product manufactured in India and approved by the reference regulatory agencies, applicable to GDA of products manufactured in India)
What are the reference drug regulatory agencies recognized by Singapore?
How does your regulatory strategy support successful product registration in Singapore?
- Product registration strategy
- Writing for Clinical, Non-Clinical, and Quality modules
- Dossier Gap Analysis
- Dossier Preparation and Submission
- Pre-Submission meetings with Health Authority (HSA, Singapore)
- Response to Health Authority Queries
- Obtaining Successful approval and licenses
What will be the important points to consider during the pre-submission preparation for Singapore?
- Identify the application type
- Decide the evaluation route to choose (can be decided with registration guides later)
- Choose the application dossier format (can be decided with registration guides later)
What are the conditions to decide the appropriate type of NDA application for my biological product?
| Application Type | Conditions |
|---|---|
| NDA-1 | For the first strength of a product containing a New Biological Entity |
| NDA-2 | For the first strength of a product containing: |
| New combination of registered biological entities | |
Registered biological entities in either:
|
|
| Registered biological entities with new route of administration | |
| For products not applicable under NDA-1 or NDA-3 | |
| NDA-3 | For subsequent strengths of a product registered or submitted as NDA-1 or NDA-2. Product name, dosage form, presentation, indication, dosing regimen, and patient population should be the same as NDA-1 or NDA-2. |
Regulatory Expertise Across
Multiple Countries












Blogs

Cell and Gene Therapy
Cell and Gene Therapy Products are modern treatments to treat diseases. Treatment of these...

Cosmetics Regulations & Registration
Germany is one of the biggest cosmetics markets in Europe, but entering the market...

Why Medical Device Submissions
Introducing a new medical device to the international market requires that you not only...


