- Home
- >
- Singapore
- >
- Medtech
Medical Device Regulatory Services in Singapore
Get expert help with medical device approvals, licensing, and regulatory compliance in Singapore. Trusted by global MedTech brands.
Medical Devices Compliance Services in Singapore
Getting your medical device approved in Singapore means working closely with the HSA—and that process can be demanding. At Artixio, we help you handle the paperwork, timelines, and compliance steps so you can focus on getting your product to market faster.
The Health Sciences Authority (HSA) manages all medical device regulations in Singapore, from import and manufacture to supply and export. We work alongside your team to make sure everything meets their standards—without unnecessary delays.
Services
Key Regulations for Medical Devices in Singapore
Classification of Medical devices
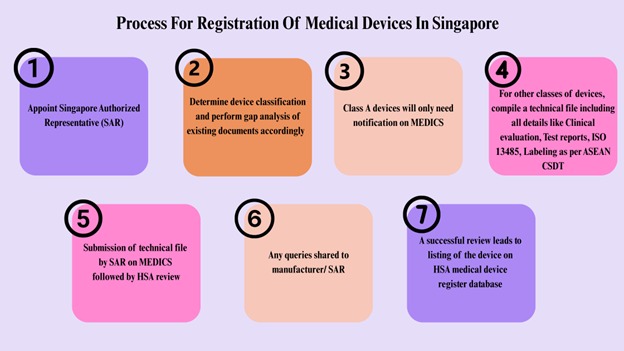
Process for Registration of Medical Devices in Singapore
Required Documents for Medical Device Registration in Singapore
- Executive Summary
- Device Description
- Design Verification & Validation
- Clinical Evidence
- Device Labelling
- Risk Analysis
- Manufacturer Information
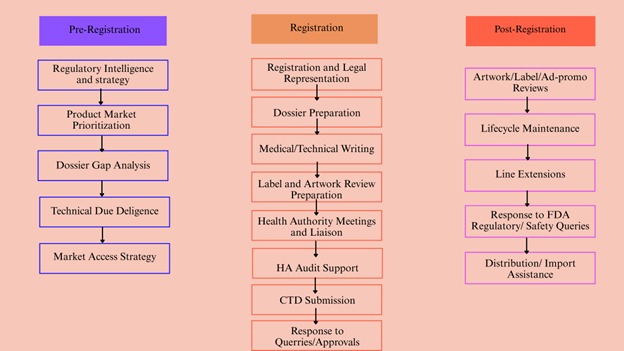
Artixio’s Medical Devices Registration Services in Singapore
Why Artixio For Medical device Consultation In Singapore?










FAQs
When is a Change Notification (CN) required?
- Technical changes to Class C and D medical devices.
- Review changes for Class B medical devices.
- Administrative changes that affect the Singapore Medical Device Register (SMDR) listing information.
- Notification changes
What are the types of medical device dealer's license issued by HSA?
What is Special access route of Medical Devices registration in Singapore?
What is Priority Review Scheme?
Regulatory Expertise Across
Multiple Countries












Blogs

Cell and Gene Therapy
Cell and Gene Therapy Products are modern treatments to treat diseases. Treatment of these...

Cosmetics Regulations & Registration
Germany is one of the biggest cosmetics markets in Europe, but entering the market...

Why Medical Device Submissions
Introducing a new medical device to the international market requires that you not only...











