- Home
- >
- South Korea
- >
- Medtech
Medical Device Regulatory Services in South Korea
We help you register and launch medical devices in South Korea with MFDS regulatory expertise and reliable local support. Work with Artixio to manage South Korea’s MFDS device approvals and stay compliant with evolving regulatory standards.
Medical Devices Regulatory Solutions in South Korea
At Artixio, we simplify MFDS approvals in South Korea—guiding medical device firms through registration, testing requirements, and regulatory liaison support.
South Korea Medical Device Regulatory Services
Key Regulations for Medical Devices in South Korea
Classification of Medical devices
Medical Devices Registration Process in South Korea
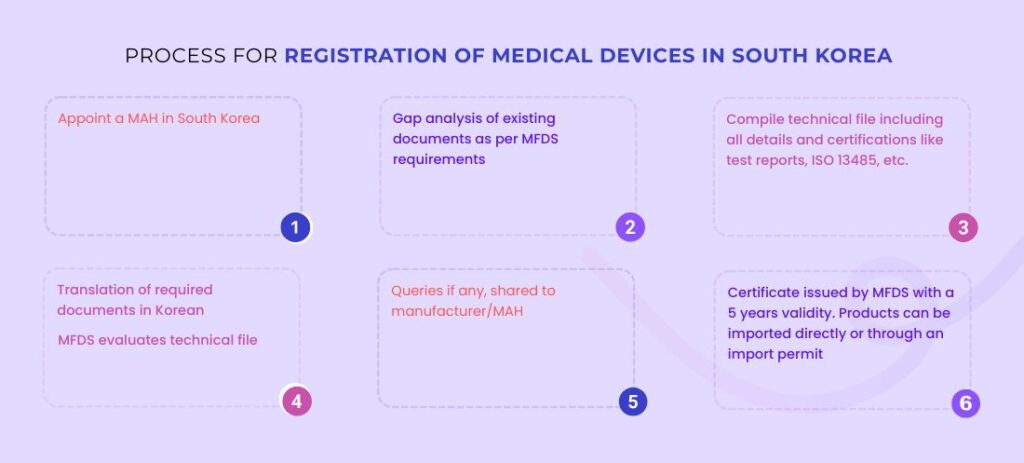
Documents Required for Registration of Medical Devices in South Korea
Administrative Documents:
- Application Form
- Supplementary Evidence
Technical Documentation:
- Intended Use
- Mechanism of Action (MoA)
- Operational (Functional) Structure
- Raw Materials
- Instructions for Use
- Test Specifications
- Clinical Trial Data (if applicable)
Medical Device Registration & Regulatory Services in South Korea
Our Regulatory and Market Access experts based in South Korea, coupled with the Global Regulatory Projects team, have helped over a hundred pharmaceutical, medical devices, diagnostics, cosmetics, nutraceuticals, supplements, and veterinary products successfully register and maintain compliance in South Korea. Combined with Artixio’s intelligence-based services platform, Dvarka, innovators and manufacturers from around the world have benefited by seamlessly managing the regulatory strategy across the product development and commercialization value chain.
From strategy and registration to post-approval support, Artixio helps medical device companies navigate South Korea’s MFDS requirements with reliable, practical regulatory guidance.
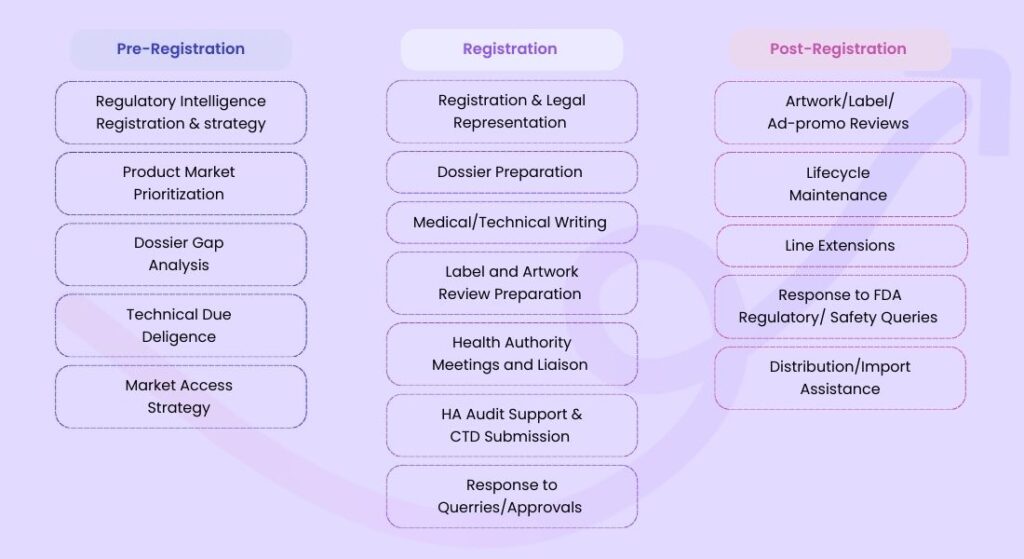
Why Choose Artixio for Medical Device Approval in Singapore?










FAQs
What is the requirement for quality management system (QMS) in South Korea?
Doe the local representative for medical device registration in South Korea is required for foreign manufacturer?
What is the validity of Medical Devices registration in South Korea?
What are the requirements to be a license holder in south Korea?
Related South Korea Services
Regulatory Expertise Across
Multiple Countries












Blogs

Cosmetics Regulations & Registration
Failure to fulfil the regulatory requirements will lead the cosmetic product to be rejected,...

Cell and Gene Therapy
Cell and Gene Therapy Products are modern treatments to treat diseases. Treatment of these...

Cosmetics Regulations & Registration
Germany is one of the biggest cosmetics markets in Europe, but entering the market...




















