Cultivated meat is changing the future of food. As the global population grows and meat demand rises, traditional livestock farming faces increasing challenges. Using advanced technology, cultivated meat provides a sustainable alternative—real meat produced without raising or slaughtering animals.
What Is Cultivated Meat?
Cultivated meat, also known as lab-grown meat, cell-based meat, or cultured meat, refers to meat products that are produced by in vitro cultivation of animal cells, rather than by traditional animal agriculture methods. The process typically involves taking a small sample of animal cells, such as muscle cells, and then culturing and multiplying them in a controlled environment, such as a bioreactor, to grow into meat tissue. Cultivated meat cannot be classified as vegan or vegetarian, nor is it categorized as genetically engineered food.
This technology has gained attention due to its potential to address various concerns associated with conventional meat production, including animal welfare, environmental impact, and food security. Cultivated meat has the potential to reduce the need for large-scale animal farming, thereby lowering greenhouse gas emissions, land use, and water consumption associated with traditional livestock agriculture.
While cultivated meat has shown promise, there are still challenges to overcome, such as scaling up production, reducing production costs, and achieving taste and texture parity with conventional meat. However, ongoing research and development efforts are aimed at addressing these challenges, with several companies working to bring cultivated meat products to market in the coming years.
Global Market for Cultivated Meat Companies
The cultivated meat industry is experiencing significant growth, with approximately 156 companies estimated to be actively operating in this space in 2022. This count encompasses not only companies engaged in the formulation and manufacturing of cultivated meat but also those involved in supporting processes such as bioreactors, cell culture media, and optimization ingredients. The majority of these companies are concentrated in countries like the USA, Israel, UK, and Singapore, reflecting the global interest and investment in this innovative sector.
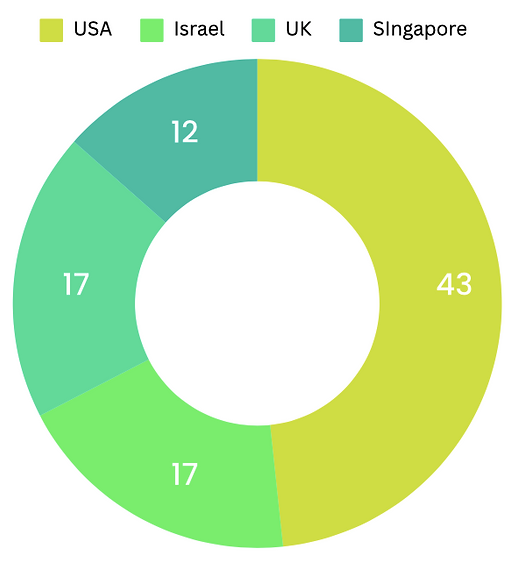
Advancements and Regulatory Milestones in Global Cultivated Meat Market
The global landscape of cultivated meat products is rapidly evolving, with several key milestones marking its progress. Singapore made history in December 2020 by approving the first cultivated chicken developed by US-based Good Meat, followed by the USA granting approval to two cultivated chicken products in June 2023. Israel also joined the ranks by approving the cultivated beef produced by Aleph Farms in January 2024.
Aleph Farms also made progress by submitting a regulatory application to the Swiss Federal Food Safety and Veterinary Office (FSVO) in July 2023, aiming to sell their Aleph Cuts in Switzerland, in collaboration with Migros, Switzerland’s largest food enterprise. These developments underscore the growing acceptance and regulatory advancements in the cultivated meat industry on a global scale. In September 2023, The Cultivated B (TCB) initiated discussions with the European Food Safety Authority (EFSA) to seek novel food approval for a cultivated meat sausage product, marking a significant step forward in the European market.
In the United States, the FDA completed its first premarket consultation with UPSIDE Foods in November 2022, followed by another consultation with GOOD Meat in March 2023, both for cell-cultivated chicken products. Subsequently, in June 2023, the USDA’s FSIS issued grants of inspection to UPSIDE Foods and GOOD Meat, approving their manufacturing facilities for cell-cultivated meat production. Notably, on July 1, 2023, UPSIDE Foods and GOOD Meat made history by selling their first cell-cultivated chicken products in the United States.
Global Regulations for Cultivated Meat Products
The global regulation of cultivated meat products is still evolving, with various countries and regions taking steps to develop frameworks to govern the production, safety, and labeling of these innovative food products. Cultivated meat products have not yet been approved for commercial sale and consumption on a global scale. However, several countries and regions are actively working on regulatory frameworks to assess the safety and suitability of cultivated meat for public consumption. Here are some updates on the regulatory status of cultivated meat in various regions:
Singapore: In December 2020, Singapore became the first country to approve the sale of cultivated meat. The Singapore Food Agency granted regulatory approval to San Francisco-based company Eat Just for its lab-grown chicken product, which was subsequently introduced to the market in limited quantities.
Israel: Israel has also made significant strides in regulating cultivated meat. The Israeli government has been supportive of cultivated meat research and development, and several Israeli companies are at the forefront of this emerging industry.
Europe: The European Union (EU) has initiated discussions on the regulatory framework for cultivated meat. In December 2020, the European Parliament passed a resolution calling for the development of clear regulations for cultivated meat products. The European Food Safety Authority (EFSA) is actively studying the safety and nutritional aspects of cultivated meat.
United States: In the United States, the regulatory landscape for cultivated meat is overseen by the Food and Drug Administration (FDA) and the U.S. Department of Agriculture (USDA). Both agencies have been working with cultivated meat companies to establish regulatory guidelines for product safety and labeling.
Other Countries: Several other countries, including Japan, South Korea, and Australia, are also exploring the regulatory framework for cultivated meat. While no commercial approvals have been granted yet, these countries are closely monitoring developments in the industry.
It’s essential to note that the regulatory approval process for cultivated meat involves rigorous evaluation of safety, quality, and labeling standards to ensure consumer protection. As the industry continues to evolve and demonstrate advancements in technology and safety, we can expect to see more countries approving cultivated meat products for commercialization in the future.
Way forward in regulation of cultivated meat
As the cultivated meat industry continues to grow and mature, regulatory agencies will play a vital role in safeguarding public health and promoting the responsible development of this emerging food technology. Here are the key considerations for regulating the cultivated meat –
Safety and Quality Standards: Regulatory agencies are focused on ensuring that cultivated meat products meet stringent safety and quality standards. This includes assessing the risk of microbial contamination, ensuring proper sanitation practices during production, and evaluating the nutritional composition of cultivated meat compared to traditional meat products.
Labeling Requirements: Clear and accurate labeling is crucial to inform consumers about the nature of cultivated meat products. Regulatory authorities are working to establish guidelines for labeling cultivated meat, including requirements for product names, ingredient lists, and nutritional information. This helps consumers make informed choices and distinguishes cultivated meat from conventional meat products.
Approval Process: Countries are developing procedures for the approval and regulation of cultivated meat products. This may involve pre-market safety assessments, inspections of production facilities, and ongoing monitoring to ensure compliance with regulatory standards. Regulatory agencies collaborate with cultivated meat companies to establish transparent and science-based approval processes.
International Collaboration: Given the global nature of the cultivated meat industry, international collaboration is essential to harmonize regulatory approaches and facilitate trade. Regulatory agencies participate in forums such as the Codex Alimentarius Commission, which develops international food standards, guidelines, and codes of practice. Collaborative efforts help establish common principles for the regulation of cultivated meat products across borders.
Public Engagement and Transparency: Regulatory authorities engage with stakeholders, including industry representatives, consumer groups, and scientific experts, to gather input and address concerns related to cultivated meat regulation. Transparency in the regulatory process builds trust and confidence in cultivated meat products among consumers and stakeholders.
While progress has been made in developing regulatory frameworks for cultivated meat, challenges remain, including the need to address scientific uncertainties, establish consensus on labeling requirements, and ensure effective enforcement of regulations.
As we stand on the cusp of a new era in food technology, cultivated meat represents a groundbreaking innovation with the potential to revolutionize the way we produce and consume meat. By offering a sustainable, ethical, and environmentally friendly alternative to conventional meat, cultivated meat holds the promise of addressing some of the most pressing challenges facing our food system. While there are still hurdles to overcome, the momentum behind cultivated meat continues to build, fueled by a shared vision of a more sustainable and humane future. As research and development efforts progress, cultivated meat may soon transition from a concept on the cutting edge of science to a staple on our dinner plates, shaping the future of food for generations to come.
Learn more about the regulatory landscape and how Artixio can assist your cultivated meat ventures! Reach out to us today to explore our capabilities and expertise in navigating global regulations.
References –
- https://crsreports.congress.gov/product/pdf/R/R47697
- https://www.thecultivatedb.com/the-cultivated-b-initiated-pre-submission-process-towards-efsa-certification-for-cultivated-sausage/




