Selling a health supplement in Singapore doesn’t involve a registration certificate. There’s no approval letter to wait for. But the regulatory compliance checks still exists. Ingredient suitability, claim wording, and safety thresholds must be checked carefully. If something goes wrong, accountability sits with the business.
Below is a brief overview of how health supplements are defined, what requirements apply, and what to review before market supply.
Defining Health Supplements in Singapore
A health supplement, as per Singapore regulations, serves as a dietary addition supporting, enhancing, or improving bodily functions without being an injectable or sterile preparation. Items utilized for animals and those packaged as food or beverages, like biscuits, cookies, coffee, and juice, do not fall under the category of health supplements.
Ingredients and Dosage Forms
From a compliance perspective, format and composition both matter. Health supplements are expected to be supplied in measured oral forms such as tablets, capsules, softgels, powders, or liquids. Health supplements ingredients fall within the range of nutrients or other substances associated with supporting normal body functions, rather than therapeutic treatment. Products intended for injection or containing medicinal components would fall outside this scope.
HSA Health Supplements Registration Steps in Singapore
In Singapore, health supplements are placed on the market without prior product approval. The responsibility sits with the company selling or importing the product. Following steps reflect how regulatory affairs in Singapore typically manage health supplement market entry.
1. Decide the product category
Before anything else, confirm the product is a health supplement and not something that would fall under medicinal control.
2. Go through the formula carefully
Check each ingredient. Make sure nothing restricted is included and that vitamin or mineral levels are reasonable.
3. Check how the product is described
Look at the wording used on the label and in marketing. It should refer to supporting normal health only. Avoid medical-type language.
4. Prepare the label
The label should clearly state:
- Product name
- Ingredients
- Directions for use
- Net content
- Name and address of the local company
- Country of origin
- Information must be in English.
5. Arrange a local importer
The business bringing the product into Singapore must be locally registered and hold the necessary food import licence.
6. Import and distribute
Submit the required import declarations and release the product for sale once compliance is ensured.
7. Keep records after launch
Maintain product documentation. If questions arise from the authorities, supporting information should be available.
Health Supplements Regulations In Singapore
Food products are regulated by the Singapore Food Agency (SFA), but health supplements come under the Health Sciences Authority (HSA). Health supplements are treated differently and they are not subject to pre-market approval by HSA. There is no licensing requirement before import or sale. Claims must stay within what is allowed for supplements. Companies can also submit a voluntary notification, though this is optional.
HSA Health Supplements Labelling Standards
In Singapore, during the health supplements registration process, most regulatory questions tend to come back to what is printed on the packaging. The label should clearly show what the product is and what it contains. Consumers should be able to see how it is meant to be taken, when it expires, and which company in Singapore is responsible for it. This information needs to appear in English.
Where companies tend to face issues is in the wording of claims. Supplements can refer to supporting normal functions, but they should not suggest medical treatment or disease prevention. Careful review of claim language helps avoid compliance concerns.
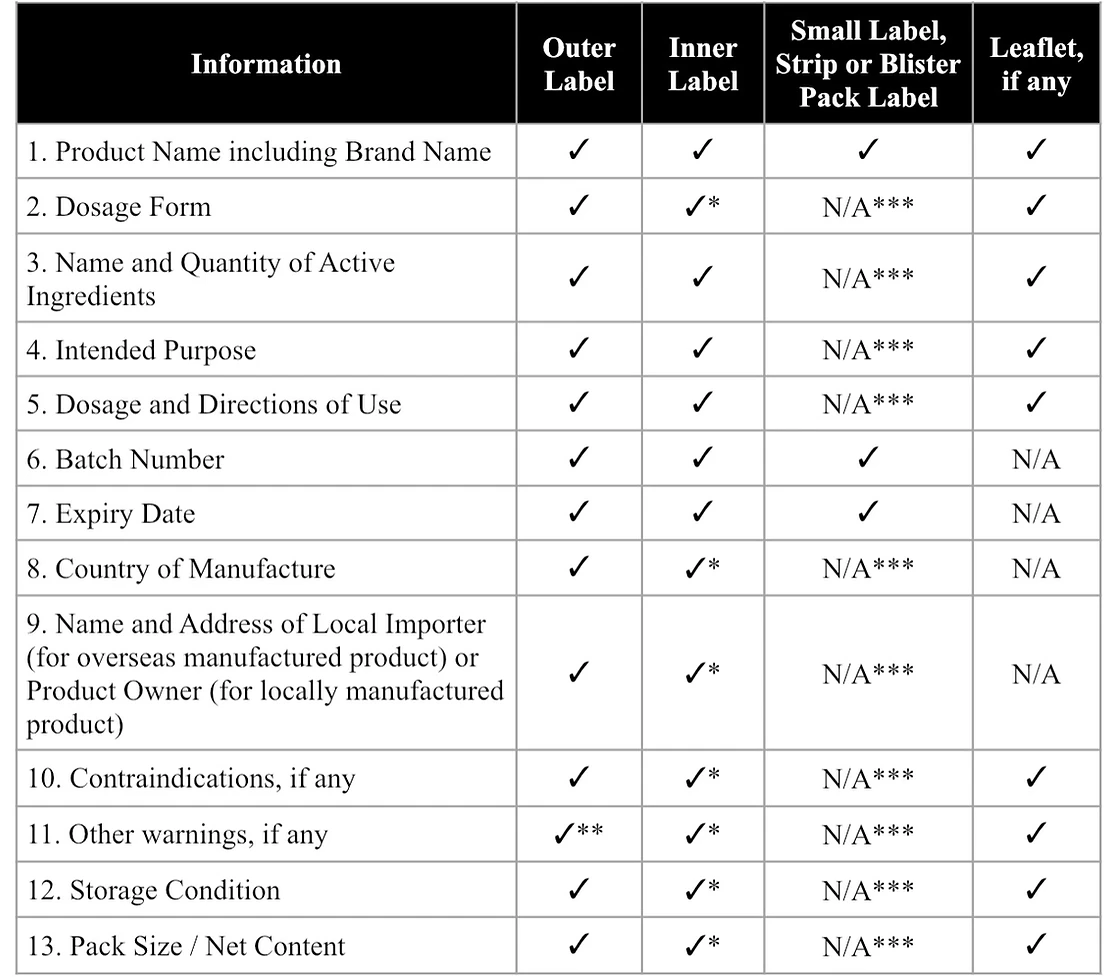
* May be omitted if the product is supplied with an outer label
** May be omitted if the accompanying product leaflet provides this information
*** The small label, strip or blister pack label should minimally display the product name, batch number and expiry date.
Safety Measures by HSA
Certain boundaries are clear. Medicinal ingredients — including substances such as steroids — are not permitted in health supplements. Limits also apply to heavy metals and other contaminants. Importers, manufacturers, wholesalers, and sellers bear the responsibility of guaranteeing product safety before supplying health supplements in Singapore.
Health Supplements Claims and Advertisements
Health supplement claims are statements or representations made regarding a product’s benefits, actions, or indications, found in various materials such as product labels, advertisements, point-of-sale materials, and brochures.
These claims fall into two categories:
- General claims
- Specific claims
Health supplements can describe how they support normal body functions. For example, wording such as “supports joint health” or “helps maintain energy levels” is generally acceptable because it refers to normal conditions. What should be avoided is any suggestion that the product treats, cures, or prevents a disease. Even indirect references to medical conditions can create regulatory issues. This applies not only to the label, but also to advertisements, brochures, websites, and social media posts. If the message sounds like a medical promise, it is likely outside what is allowed for a supplement.
Voluntary Notification Process for Health Supplements
Only locally registered companies under the Accounting and Corporate Regulatory Authority (ACRA) are eligible to submit product notifications. There are no fees associated with the submission of a product notification. CorpPass authentication is necessary for the submission process. The submission requires a complete notification form, accompanying declarations, and documents attesting that the product meets the prescribed quality and safety standards.
To complete the notification process, various documents must be submitted to HSA_TMHS@hsa.gov.sg, including a copy of the form, manufacturer’s license, certificate of analysis, product label, product leaflet, laboratory test reports, and relevant undertaking forms. Detailed guidelines for testing requirements and a checklist for submission are provided by HSA for reference.
Upon submission, the turn-around-time for processing new product notifications is 60 working days. This timeline excludes the duration required by the company to respond to any requests for additional information or clarifications (company’s stop-clock).
HSA reserves the right to refuse notifications for products with non-compliance records or those under HSA’s investigation. Products failing to meet notification conditions risk removal from the list of notified products.
Conclusion
Getting a health supplement ready for sale in Singapore usually looks simple on paper. In practice, most issues come from small details — an ingredient level that was not double-checked, a claim that reads too strongly, or a label missing required information. These are the points that tend to slow things down later. Reviewing everything properly before import helps avoid unnecessary back-and-forth.
If you are working on a health supplement regulatory and registration process in Singapore and want someone to go through the formulation or packaging with you, we can help. You can write to us at info@artixio.com to discuss about your health supplements regulatory process.




