For cosmetic brands and regulatory teams, Mexico often looks straightforward, as there’s no pre-market registration. In reality, delays happen at the import. Shipments are frequently held due to NOM-141 labeling errors, misclassification, or missing importer details. This is one of the most common causes of customs detention, often leading to returns or destruction.
The challenge is that compliance is enforced late, either at customs or post-market, which means teams often discover gaps when products are already in transit. Even without registration, every product must meet COFEPRIS requirements on labeling, ingredients, GMP, and local representation, and missing any one of these can block market entry.
If you’re planning an entry, understanding where these breakdowns actually happen is critical. This blog breaks down exactly how to avoid these delays step by step.
Cosmetic Classification in Mexico
In order to comply, you must make sure that your products are classified as cosmetics.
In Mexico, the cosmetics category is limited to the products that are directly applied to the external body parts, including skin, hair, lips, or nails, to clean or beautify them.
Products may fall under the drug category under COFEPRIS cosmetic regulations if:
- Claim to treat or prevent conditions such as acne or pigmentation
- Affect the structure or function of the skin
- Contain ingredients associated with pharmacological action
Who Regulates Cosmetics in Mexico?
In Mexico, cosmetics are controlled by COFEPRIS, the national health authority of the Ministry of Health.
It oversees key aspects of cosmetic compliance, including:
- Manufacturing and quality standards
- Import and commercialization
- Labeling and product claims
- Post-market inspections and enforcement
The regulatory framework is governed through:
- General Health Law
- Regulation on Sanitary Control of Products and Services
- Applicable NOM standards (such as labeling and GMP)
Regulations Checklist for Cosmetics in Mexico
Cosmetic compliance in Mexico depends on meeting different requirements regulated by COFEPRIS. Below is a brief checklist to help you get started:
| Requirement | What to Ensure |
| Legal Framework |
Compliance with General Health Law and Regulation on Sanitary Control of Products and Services
|
| Labeling (NOM-141) | Spanish label including:
• Product name and function • Full ingredient list (INCI) • Net content • Manufacturer & local importer details • Country of origin • Instructions for use |
| GMP (NOM-259) |
Manufacturing meets quality and safety standards (applies to imported products as well)
|
| Ingredient Compliance | No restricted or prohibited substances in formulation |
| Notice of Operation |
Filed by local manufacturer/importer/distributor before starting operations
|
| Local Representation | Mexico-based importer or legal entity in place |
| Import Readiness | Documentation and labeling aligned for customs clearance |
| Post-Market Oversight |
Prepared for inspections, product checks, and enforcement
|
Documentation Required for Compliance in Mexico
Companies must keep good records, particularly of the Notice of Operation and inspections, to remain in compliance.
Typical documents include:
- Notice of Operation form
- Business registration and tax documents
- Proof of address
- Identification of the legal representative
- Company incorporation documents
- Details of the responsible professional
- Product formulation and technical documentation
- Manufacturing and quality documentation
- Proof of payment of fees
With a well-structured Product Information File, audits become a lot easier to manage.
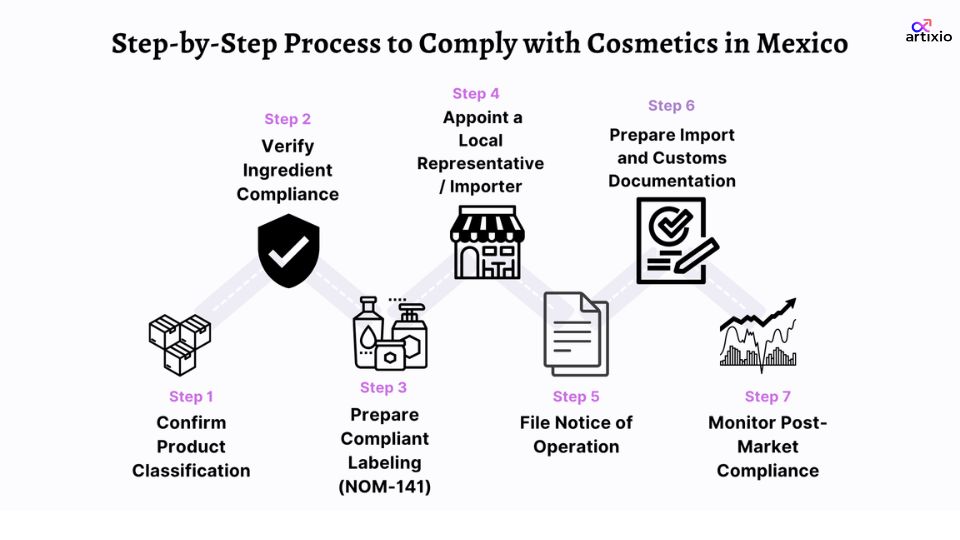
Step-by-Step Cosmetics Registration Process in Mexico
It helps to follow a systematic compliance route before legally entering the cosmetics market in Mexico. Below are the steps:
Step 1: Confirm Product Classification
Confirm that your product is cosmetic in terms of formulation and claims. When it is a therapeutic category, it will have to take a different regulatory route and approval process.
Step 2: Verify Ingredient Compliance
Compare the formulation with the allowed and prohibited substance lists in Mexico. Any ingredient that is non-compliant will have to be reformulated prior to entering the market.
Step 3: Prepare Compliant Labeling (NOM-141)
Create Spanish labels with all the required information, such as ingredients (INCI), importer, usage instructions, and warnings. One of the most frequent reasons for delays in imports is label issues.
Step 4: Appoint a Local Representative / Importer
Foreign firms are required to conduct business through a local entity based in Mexico. This partner handles regulatory submissions, import clearance, and communication with COFEPRIS.
Step 5: File Notice of Operation
The local entity has to file the Notice of Operation prior to commencing any manufacturing, importation, or distribution operations. This facilitates legal business in Mexico.
Step 6: Prepare Import and Customs Documentation
Make sure that all the documentation, labeling, and declarations are in place prior to shipment. Any discrepancy may lead to customs detentions or rejection.
Step 7: Monitor Post-Market Compliance
Once launched, keep product records, observe safety, and be ready to be inspected. COFEPRIS implements compliance by conducting continuous surveillance in the market.
Regulatory Requirements for Cosmetics in Mexico
Companies need to meet key compliance requirements set by COFEPRIS. These include labeling, ingredient safety, manufacturing standards, and import rules.
- Labeling Requirements (NOM-141)
Labeling is where many companies either succeed or fail. Cosmetic labels must include essential details such as ingredients, net quantity, manufacturer information, and instructions for use. All information must be in Spanish, clear, and accurate.
In practice, labeling errors are a major cause of customs delays. Even small issues, such as incomplete translations, can lead to products being held or rejected. This is why experienced companies treat labeling as a critical step rather than a final task when planning Mexico cosmetics import requirements.
- Good Manufacturing Practices (GMP) – NOM-259
Good Manufacturing Practices are used to control the quality of products. This includes the manufacturing process of products, the maintenance of facilities, employee training, and quality inspection. These standards are not only applicable to manufacturers in Mexico but also abroad. Simply put, the authorities like to be assured that your product is produced in a consistent and safe manner.
- Notice of Operation (Aviso de Funcionamiento)
Companies are not required to register every product, but they are required to file a Notice of Operation prior to starting business. This is applicable to manufacturers, importers, distributors, and even storage facilities.
It informs regulators that you are in business and that you are operating within the regulations. It does not endorse your products; it just enables your business to run within the law. You cannot sell in Mexico without it. For foreign companies, this typically involves using a local representative.
- Ingredient and Safety Compliance
It is the full responsibility of companies to ensure the safety of their products. The ingredients should be in accordance with Mexican laws, such as the prohibition of some substances. In some cases, acceptable products in other countries require modifications to suit local regulations.
It is best to maintain a Product Information File since it will assist you in being ready in case the authorities demand information about your product.
- Importation and Local Representation Requirements
Mexico does not allow foreign companies to operate independently. It needs a local representative or importer, and this partner takes care of communication with regulators, filing documents, and compliance.
They also play a key role during imports, where labeling and paperwork are checked. It is not feasible to enter the Mexican market without a local presence.
- Advertising and Claims Compliance
In Mexico, cosmetic claims are highly controlled to avoid misleading or therapeutic claims. Unless registered as drugs, products should not purport to diagnose, treat, or prevent medical conditions. The claims should be factual, supported, and consistent with the product being a cosmetic.
COFEPRIS controls advertising and promotion materials, and the breach may lead to fines or removal of the product from the market.
Conclusion
Although Mexico does not require traditional cosmetic registration, the regulatory burden is high. Firms should have a clear understanding of classification, labeling, GMP, local representation, and post-market requirements. Registration is substituted by compliance in this environment as the most important barrier to entry.
If you need assistance with your regulatory journey while entering the Mexican market, experts at Artixio provide Cosmetics Regulatory support in Mexico. Our team helps you ensure that your products are compliant with COFEPRIS regulations and are ready to enter Mexico. Get in touch with us at info@artixio.com.
FAQs
1. Are imported cosmetics required to be registered in Mexico?
No, the majority of cosmetics do not have to be registered, but they have to comply with all labeling, ingredient, and importation requirements.
2. Can I sell cosmetics in Mexico without a local entity?
No, foreign firms have to work with a Mexico-based importer or representative who will work with COFEPRIS.
3. Why do shipments of cosmetics get detained at Mexican customs?
Normally, it is caused by labeling mistakes, wrong classification, incomplete importer information, or a lack of documentation.
4. Do we have any special types of cosmetics that need to be approved in Mexico?
Yes, therapeutic products can be considered drugs and must be sanitarily registered.
5. What is the time taken to introduce a cosmetic product in Mexico?
No set time frame; labeling problems, non-conformance of ingredients, or local set-up often cause delays.